Barth syndrome: A potential treatment for a rare disease
Barth syndrome is a rare disorder in males caused by a variant of the gene TAFAZZIN. It affects the metabolism of the fat molecule cardiolipin in mitochondria, resulting in the dysfunction of skeletal muscle and the heart. The syndrome doesn’t have a specific therapy, so patients who suffer from it have health problems their entire lives and are more likely to die as a result of heart problems. Dr Hilary J Vernon at Johns Hopkins University in the USA studied the efficacy and safety of elamipretide, a protein that targets the mitochondria and interacts with cardiolipin – with promising results.
Barth syndrome is a rare, serious genetic disorder that affects one in 1,000,000 males. It is caused by an alteration of the gene TAFAZZIN within the X chromosome. This gene is responsible for the synthesis and structure of cardiolipin (CL), a fat molecule essential to the function of mitochondria, the energy factory of our cells.
CL is not only important for the structure of mitochondria but also for its function, including the capacity to transport proteins and to create energy. When TAFAZZIN is mutated, immature CL species accumulate, leading to the increased ratio of monolysocardiolipin (MLCL) to CL that is diagnostic for patients with Barth syndrome. This ultimately leads to mitochondrial dysfunction and such sequelae as weakening of the skeletal muscles and the heart.
Living with Barth syndrome
Barth syndrome is a multisystemic disorder, which means it affects many systems in the body. Since problems in energy production inside the cells affect mainly the heart and the skeletal muscles, signs and symptoms include a weakened heart (cardiomyopathy), muscle weakness (myopathy), fatigue, exercise intolerance, motor skills delay, and growth problems. The disorder can lead to heart failure and heart transplantation before adulthood.
Symptoms of Barth syndrome have a negative social and emotional impact, especially in children and teenagers who aren’t able to engage in physical activities.Another characteristic of Barth syndrome is neutropenia (ie, low levels of white blood cells), which makes patients with Barth syndrome more vulnerable to infections. The mortality in Barth syndrome is high, and the symptoms have a negative social and emotional impact, especially in children and teenagers who are unable to play, engage in physical activities, or participate in sports.
A potential treatment?
Currently, no curative therapies for the treatment of patients with Barth syndrome exist and supportive care is of limited clinical impact. Dr Hilary J Vernon, Associate Professor of Genetic Medicine at the Johns Hopkins University and attending physician at the Kennedy Krieger Institute, studied the efficacy and safety of elamipretide, a potential treatment for Barth syndrome. Vernon and her colleagues oversaw TAZPOWER, a randomised, double-blind, placebo crossover trial that assessed the safety and efficacy (ie, effectiveness) of elamipretide in Barth syndrome patients.
Elamipretide is a peptide that migrates inside the cells and targets the mitochondria. It travels to the inner wall of mitochondria and binds to CL, thereby improving the stability of the mitochondrial structure, enhancing the production of ATP (ie, energy), and reducing the production of free radicals. In preclinical models, that is, in cell cultures and animals, elamipretide has been shown to improve the structure and function of mitochondria, leading to improved heart and skeletal muscle function.
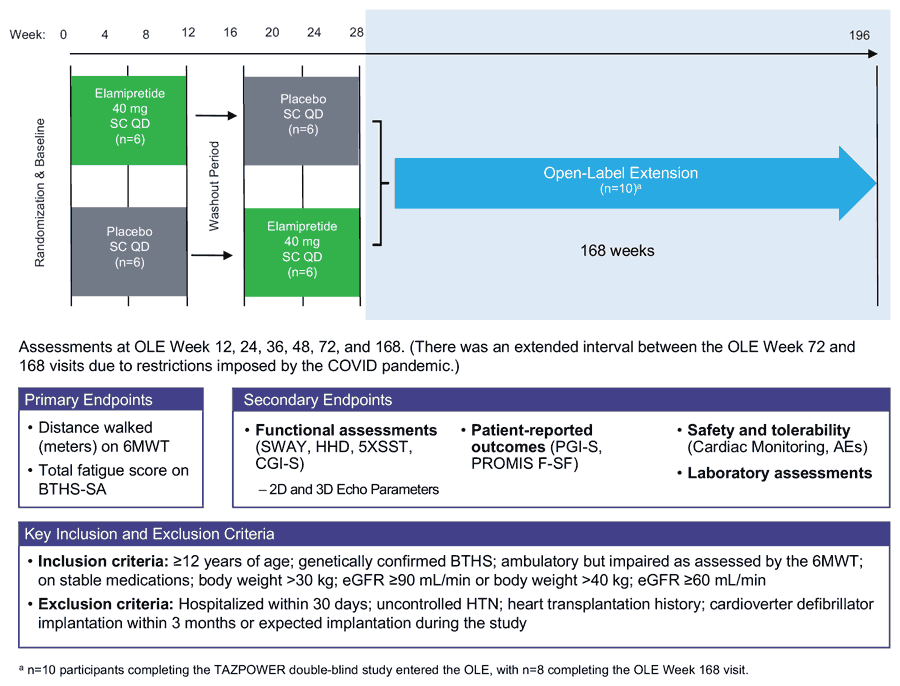
The TAZPOWER trial
In the TAZPOWER trial and its open-label extension (OLE), Vernon and her team tested the long-term safety, efficacy, and tolerability of elamipretide. To test the efficacy of elamipretide, the researchers measured the skeletal muscle function of the participants, the ratio of CL and MLCL, the presence and severity of symptoms through different physician and patient outcomes scales, and the cardiac function as assessed with echocardiography.
Researchers evaluated the exercise capacity of muscles, muscle strength, balance, and fatigue through the following tests: the six-minute walk test (6MWT), the Barth syndrome symptom assessment (BTHS-SA), the muscle strength by hand-held dynamometry (HHD), the five times sit-to-stand test (5XSST), and the SWAY balance test.

A total of 12 male participants, diagnosed with Barth syndrome and at least 12 years of age (average age 19 years), were included in the TAZPOWER trial. For 12 weeks, six of the participants received a daily subcutaneous injection of 40mg of elamipretide, and the remainder were given a placebo. Neither the participants nor the physicians knew who was receiving elamipretide. After 12 weeks, treatment and placebo were stopped for four weeks. The purpose of this part of the study, called the washout period, was to allow for the effects of treatment to disappear. That way, the researchers could compare the effect of the treatment against the placebo in the same participants.
Next, during the 12-week crossover period, the participants who had received elamipretide were given placebo and vice versa. While an independent group of experts knew who was given elamipretide or placebo before and after the crossover, once again, both the physicians and the participants were uninformed.
Elamipretide has proved to provide long-term benefits inmuscle strength and cardiac function in
Barth syndrome patients.
After a total of 28 weeks, 10 of the 12 patients who participated in the TAZPOWER trial entered the OLE. During 168 weeks, almost four years, all participants received daily injections of 40mg of elamipretide. Eight patients completed the OLE study, and three of them remained on elamipretide for an additional 92 weeks as part of the Expanded Access Program.
Safety and efficacy
As with any other treatment, patients administered elamipretide experienced side effects, with the most common ones being injection site reactions (eg, pain, itchiness, and redness). Overall, elamipretide was well tolerated and considered to have a favourable safety profile.
After almost four years of treatment, researchers saw significant improvements in cardiac and functional performance measures. For the 6MWT, researchers found that all the participants increased the distance they were able to walk in six minutes at every visit, with a mean increase from baseline of 96.1 metres at the end of the study. Statistically significant improvements in the HHD and SWAY balance tests were also observed, indicating an increase in muscle strength.
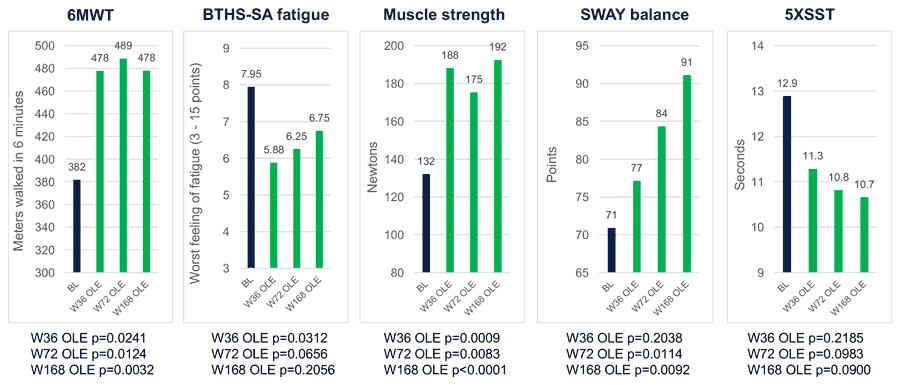
Improvements were noted in measures of symptoms as assessed by both physicians and patients. Specifically, mean improvements from baseline in BTHS-SA Total Fatigue, Patient Global Impression of Symptom Severity and Change Scale (PGI-S Q1), and Clinical Global Impression of Symptom Severity and Change Scale (CGI-S) scores were demonstrated.
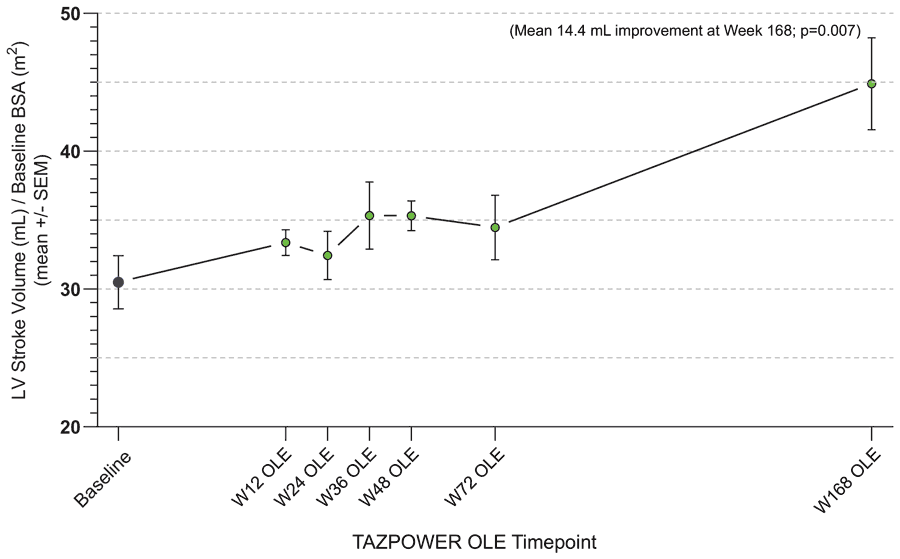
Cardiac function, as measured through echocardiography, displayed significant increases in the left ventricular stroke volume, the amount of blood pumped from the ventricle during each cardiac contraction. Statistical analysis showed that the improvement in cardiac function was related to the better muscle function seen in the 6MWT and HHD tests.
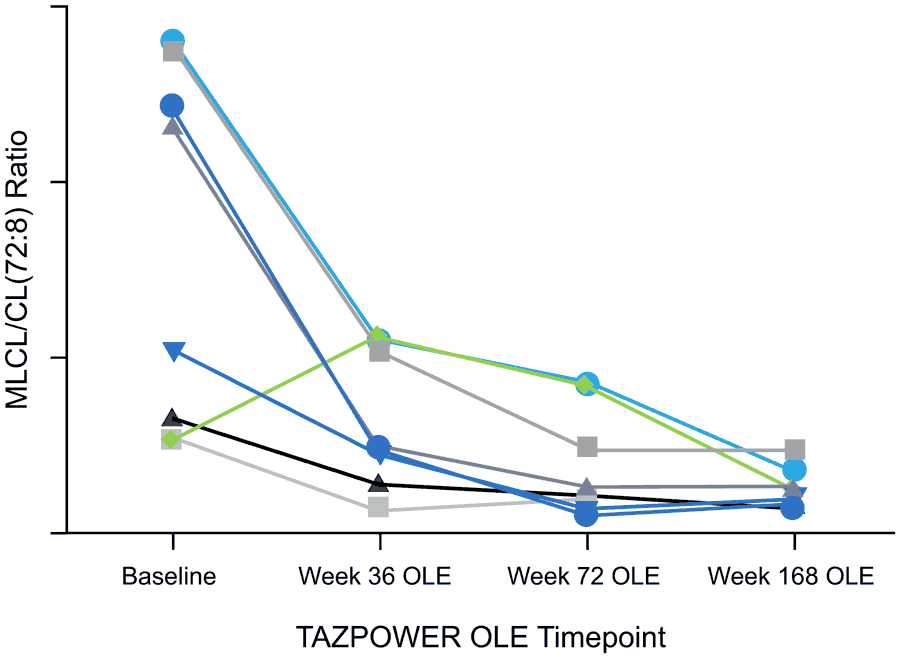
Finally, the MLCL/CL ratio decreased during elamipretide treatment, demonstrating improvement. Interestingly, researchers noted the change in the MLCL/CL ratio to be consistent with the improvement in long-term functional and cardiac measures.
In conclusion, this study provided evidence of the safety and long-term benefits in muscle strength and cardiac function in Barth syndrome patients. However, more research is needed as the number of participants was very low, given that Barth syndrome is an ultra-rare disease with a small patient pool.

Personal Response
Do you think it could be possible in the future to identify this disorder through genetic studies during pregnancy?Just as with any other genetic disorder with a known gene change, there are possibilities for prenatal diagnosis. We recommend that any family with questions in this area seek prenatal genetic counselling.
The treatment is applied through injections, do you think it could be possible to design another, less invasive format, especially for children?
The company responsible for the development of elamipretide, Stealth BioTherapeutics, recognises the importance of developing alternate forms of administration that do not include injections. I am aware of their efforts to do so, although the nature of the chemical structure of elamipretide may be compromised by the acidic environment within the GI system. But I wouldn’t be surprised if their efforts result in an alternate form of delivery someday.