Developing a novel inhaled therapeutic for the treatment of bronchiolitis obliterans syndrome
Although lung transplantation has become an established treatment option for end-stage lung disease, unfortunately extended survival for recipients remains a challenge. The primary factor impacting long-term survival following transplantation is bronchiolitis obliterans syndrome (BOS), otherwise known as obliterative bronchiolitis (OB), a devastating and fatal disease. BOS is the leading cause of death following lung transplantation, with nearly 50 percent of patients developing BOS within five years after transplantation.
Understanding bronchiolitis obliterans syndrome
Bronchiolitis obliterans syndrome is an obstructive airway disease which causes inflammation and fibrosis of the bronchiolar walls and reduces the diameter of the bronchioles. The disease can progress quickly with respiratory failure and death typically occurring one to two years after diagnosis. BOS is commonly seen in patients following lung transplantation and allogeneic hematopoietic stem cell transplant (alloHSCT), although it is also associated with autoimmune disease and exposure to environmental contaminants. Based on company market research, an estimated 21,500 lung transplant and 8,500 alloHSCT recipients are currently affected by BOS in the US, Europe and Japan.
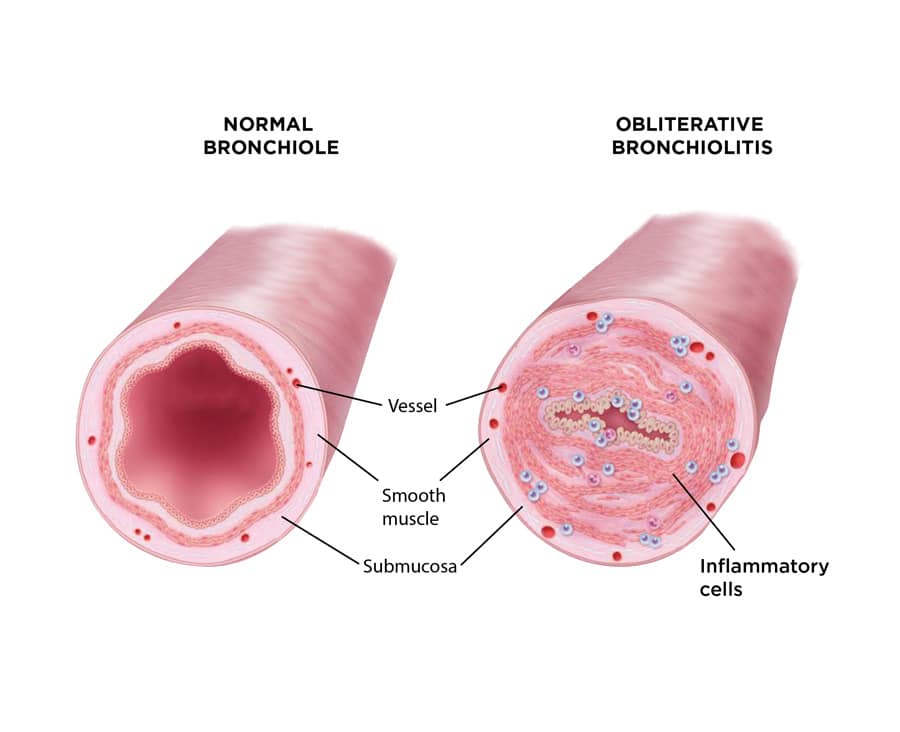
Alongside the physical consequences for patients with BOS and potential negative impact on the mental well-being of both patients with BOS and their caregivers, there is also an economic burden of the disease. US healthcare utilisation data shows a 30-40 percent annual increase in costs in treating lung transplant recipients with BOS compared to lung transplant patients without the disease. Despite this significant humanistic and economic burden, there are no approved treatments for BOS at this time.
Advancing inhaled respiratory therapy
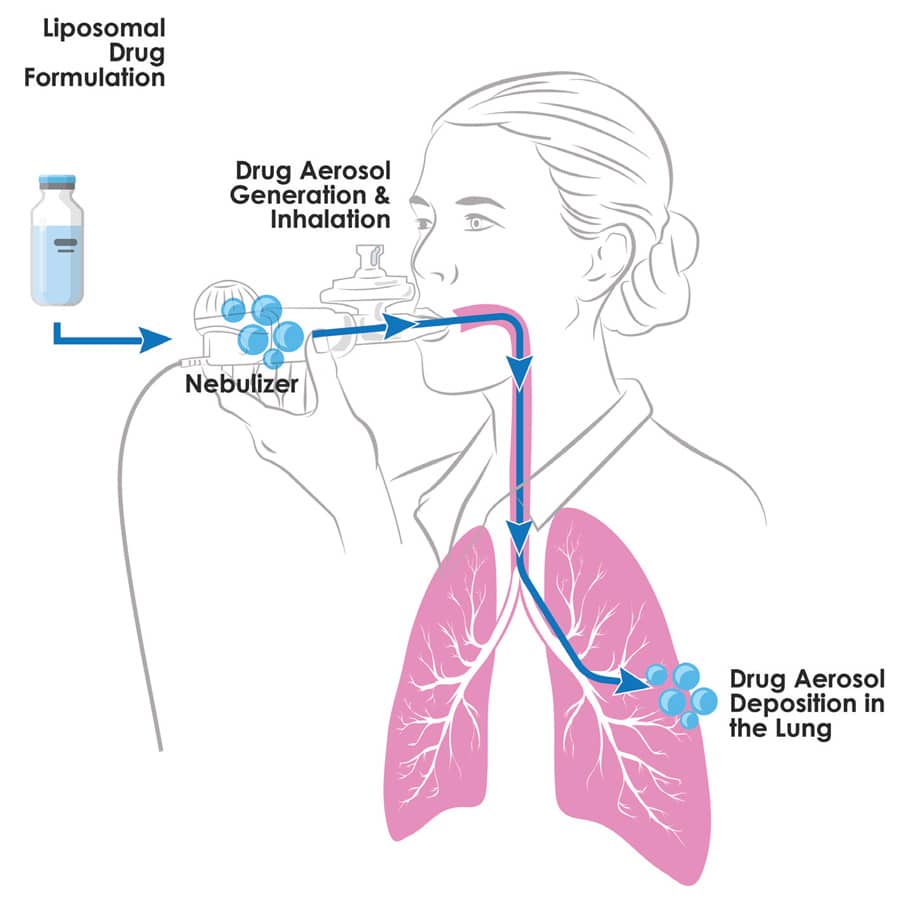
Breath Therapeutics, founded in 2016 and acquired by Zambon S.p.A. in July 2019, is led by Chief Executive Officer, Jens Stegemann, PhD and the Breath team consists of multi-skilled professionals with expertise in drug formulation and manufacturing, aerosol drug delivery, clinical development and commercialisation. The US- and EU-based company has developed a novel liposomal formulation of cyclosporine A, called L-CsA-i, which is administered directly to the small airways of the lungs via an investigational eFlow® Nebulizer System developed by PARI Pharma. The eFlow® Nebulizer System was designed specifically for use with L-CsA-i and allows drug delivery to the target area in the lung. It is designed for home-inhalation for additional convenience for patients.

L-CsA-i is currently in the experimental stages and has received orphan drug designation from the United States Food and Drug Administration and European Medicines Agency for the treatment of BOS. At time of writing, there are 1,100 patient months of L-CsA-i exposure in clinical trials. L-CsA-i is an investigational drug and its safety and efficacy have not been established.
BOS can progress quickly with respiratory failure and death typically occurring one to two years after diagnosis.
The rationale for inhalation delivery
Systemically-administered cyclosporine A (CsA) is often used following lung transplant and alloHSCT to prevent acute lung allograft rejection or acute and chronic graft versus host disease. In the systemic mode of administration at the usual target systemic concentration, CsA is not able to achieve sufficient drug levels at the site of disease in the bronchioles for the treatment of BOS. The potential advantage of inhaled therapies is to deliver a drug directly to the site of the disease and to lower systemic exposure.
L-CsA-i clinical trials for the treatment of BOS
The company is in the process of conducting five clinical trials to evaluate the safety and efficacy of L-CsA-i. The BOSTON development program aims to evaluate the use of L-CsA-i for the treatment of BOS in patients aged six and older. The BOSTON program builds upon prior research that suggests local administration of immunosuppressives in the airways may be a potential option to treat BOS.
The BOSTON-1 and BOSTON-2 clinical trials were initiated in March 2019. BOSTON-1 is evaluating L-CsA-i for the treatment of BOS
The mission of Breath Therapeutics – a Zambon Group Company is to bring the first safe and effective treatment to people with BOS.
in single lung transplant recipients whereas BOSTON-2 applies the same methodology to double lung transplant recipients with BOS. The two trials will enroll a total of 220 participants at 35 leading lung transplant specialty centers in eight countries. Upon completion of the 48-week trials, participants will be eligible to continue in BOSTON-3, an open-label extension trial of BOSTON-1 and BOSTON-2.

Two additional clinical trials are planned. The target participants for BOSTON-4 are adult recipients of alloHSCT. BOSTON-5 will study L-CsA-i in paediatric patients ages 6 to 17 years.
Breath Therapeutics – a Zambon Group Company is dedicated to developing a potential first therapy for people with BOS.
Personal Response
What are your short-term and long-term goals for this drug candidate?
At the same time, the commercial team is preparing to bring L‑CsA‑i to people with BOS upon receiving the necessary regulatory approvals in the US, Europe and other geographies. Our ultimate mission is to offer the first safe and effective treatment to people with BOS, an underserved community with an urgent unmet medical need.