Potential breakthrough in cataract treatment
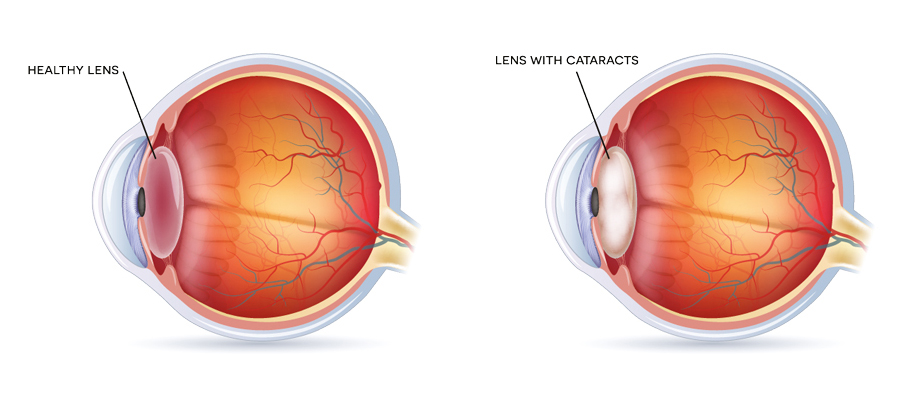
The lens of the eye is transparent and functions to focus images at various distances on the retina by changing its shape. A cataract is cloudiness or opacity in the lens, most commonly due to light scatter from aggregated lens crystallins. These are structural proteins critical to the transparency and refractivity required for lens function. When proteins in the lens become damaged, they may aggregate into light-scattering bodies, causing blurry vision, faded colours and if left untreated, eventual blindness. Cataracts are the leading cause of blindness globally, accounting for 42% of all blindness. Cataracts are most commonly caused by ageing; in developing countries they may occur at a higher prevalence and earlier age. In the USA alone, 56% of the total cost of major visual disorders amongst individuals aged 65 years or older results from cataracts. With a rise in the ageing population, cataracts will be an increasing economic and social burden.
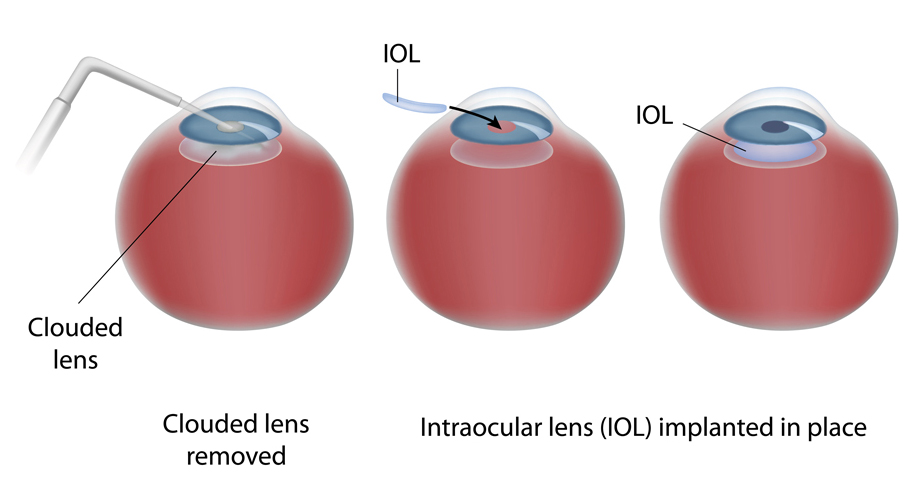
Presently, cataracts can only be effectively treated with surgery to remove the cloudy lens and replace it with an artificial lens. However, this may not restore optimum vision and cataract surgery often may not be readily available, especially in developing countries which lack adequate surgical facilities and qualified eye surgeons. Patients often need to travel long distances to obtain cataract surgery, further impacting their quality of life. In addition, cataract surgery may have complications and 30-50% of patients will need follow-up laser treatment within two years of surgery to eliminate posterior capsule opacification.
Despite these problems, surgery remains the only current method for cataract management because pharmacological interventions to date, have not been clinically shown to restore lens clarity in cataracts or to either prevent or significantly slow down cataract formation. This is very important because a 10-year delay in cataract formation would significantly reduce the incidence of cataract blindness and decrease the need for surgery by 50%. Development of non-surgical interventions to prevent or delay cataract progression would, therefore, eliminate the need for surgery for many patients.
Causes of cataracts
In addition to ageing, cataract formation is caused by other risk factors, such as smoking, diabetes, and UV light damage. Cataract is nearly always a multifactorial disease, making it difficult to develop a “general anti-cataract” drug that tackles the complex interrelationship between different risk factors. Generally, these risk factors are associated with oxidative stress which increases with age, as the natural anti-oxidative ability of the lens diminishes. Cataracts are also associated with metabolic disorders such as diabetes mellitus. These cataracts are linked to localised osmotic changes in the lens. Non-surgical interventions have been focused on reducing oxidative and osmotic stress by identifying biochemical pathways central to the formation and progression of cataract.
Oxidative stress and age-related cataracts
Oxidative stress is the result of imbalance between the rates of production and degradation of deleterious oxidants. While oxygen is essential to life, partial reduction of molecular oxygen by metabolic or photo-chemical processes produces reactive oxidative intermediates, such as free radicals and other reactive oxygen species (ROS), which are damaging to proteins and other cellular components. The presence of free transition metals, such as copper, iron, and zinc also contribute to production of ROS. The damaging effects of these oxidants accumulate in lens cells progressively as we age.
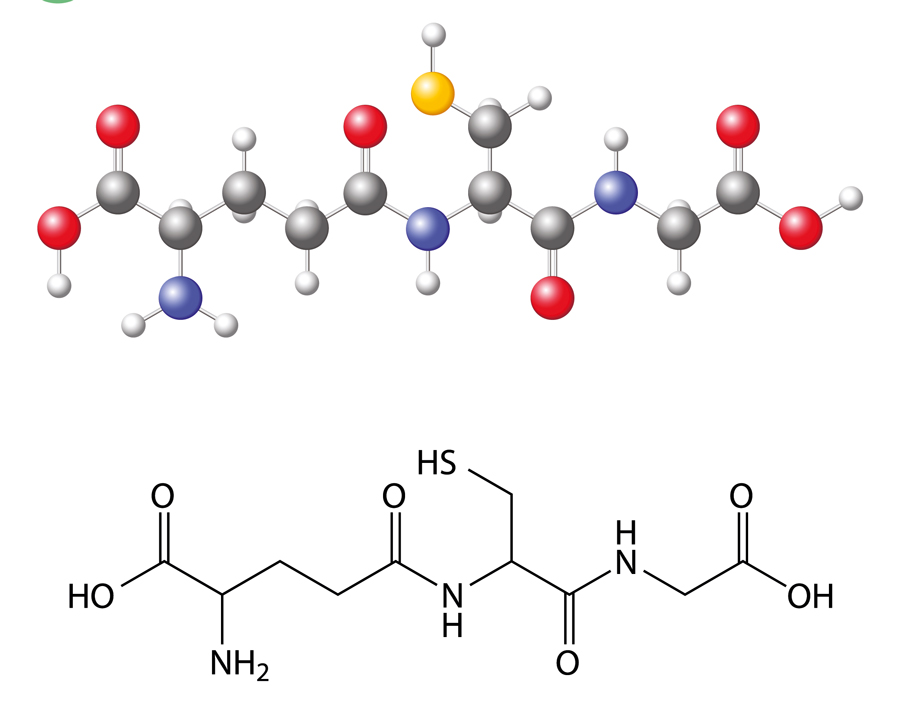
There are endogenous antioxidant defences to eliminate these oxidants and repair damaged molecules and tissues. One such antioxidant is reduced glutathione. However, these defences weaken over time through repeated exposure to dangerous exogenous risk factors such as smoking and the sun. For example, some epidemiological studies have found a positive correlation between outdoor occupations with exposure to longer and more intense daylight hours and cataract. Increasing age is also correlated with increasing oxidative stress to the lens.
Therefore, antioxidant supplements have been examined as potential anti-cataract agents. For years, researchers have studied the potential of antioxidant vitamins. Many randomised, double-blinded, placebo-controlled clinical trials with 1,000 to 40,000 participants have found inconclusive efficacy regarding antioxidant vitamins, such as vitamin C, E, and carotenoids, in preventing cataract development. These vitamins were orally administered. It has been hypothesised that oral antioxidants failed to sufficiently pass the blood-aqueous barrier and exert their therapeutic effect on the lens, as the lens receives all of its nutrients from the aqueous humour. In spite of extensive research on antioxidants and the lens, no antioxidant drug has been approved by the FDA for treatment or prevention of cataracts.
Unlike orally administered antioxidants, Optixcare® Eye Health is a topical multifunctional antioxidant that can easily reach the lens.
Metabolic cataract
Another type of cataract is called “sugar cataract”, which is common in patients with diabetes mellitus, who are up to five times more likely to develop cataracts and undergo cataract surgery at an earlier age. Sugar cataracts are assuming greater importance as diabetes continues to increase in both developing and industrialised countries. Research has shown that the sorbitol pathway is implicated in the formation of cataracts in diabetics. The enzyme aldose reductase (AR) reduces glucose to sorbitol. Normally, sorbitol is converted into fructose by the enzyme sorbitol dehydrogenase, but in diabetics, sorbitol is produced more quickly than it is being converted into fructose. The localised accumulation of sorbitol in the lens leads to hyperosmotic effects, which result in both endoplasmic stress that is linked to ROS generation in the lens epithelium and the formation of hydropic (extensive fluid accumulation) lens fibres that degenerate. Both cause cataracts.
AR, the first enzyme in the pathway, has been positively correlated genetically to the rapid onset and severity of diabetic ocular complications in diabetic patient populations globally. Therefore, researchers aim to target AR using aldose reductase inhibitors (ARIs), which can be found in natural products (nutraceuticals) such as ginger. Both nutraceutical and synthetic ARIs have demonstrated delay and prevention of cataracts in animal models and lens organ culture systems. However synthetic ARIs are more potent and more readily able to reach the lens. It has been difficult to apply these findings to human clinical trials, because the time-frame of human diabetic cataract formation has not been fully elucidated. In contrast, 75% of diabetic dogs develop cataracts requiring surgery to restore vision within the first year of the onset of diabetes. In an multi-centre placebo controlled clinical trial conducted under FDA supervision, treatment according to protocol analysis indicated that the topical ARI Kinostat® reduces the development of cataracts by 85% compared to placebo treated diabetic dogs. In a subsequent compassionate use study, Kinostat® maintained the dog’s vision for up to 8 years. The FDA has provisionally approved this drug pending completion of manufacturing based studies. By validating the premise that drugs appropriately targeted to cataract development can prevent cataract formation, Kinostat® brings a new paradigm to maintaining vision in diabetic dogs. The prevention of diabetic cataracts, especially in juvenile (Type 1) diabetics, may soon be possible.
Kinostat® brings a new paradigm to veterinary ophthalmology – it is the first and only drug that has been clinically shown to prevent vision loss from cataracts and it has received pending provisional FDA approval for marketing.
Hope for a pharmacological treatment of cataracts
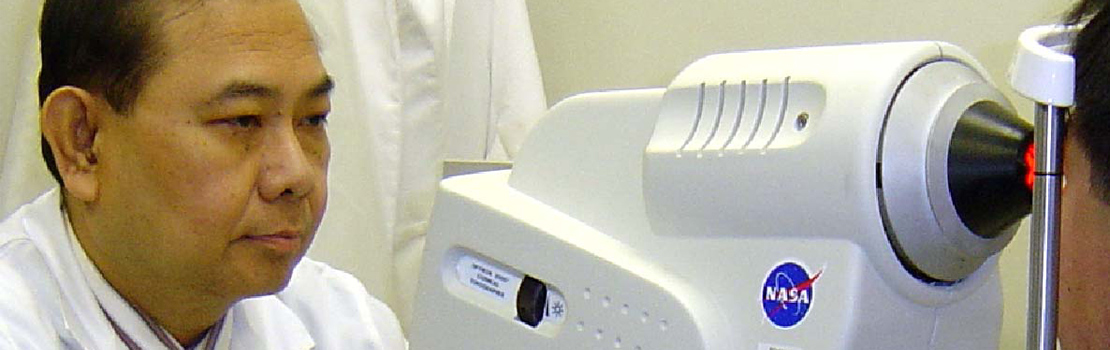
Dr Peter Kador and Dr Sam Zigler are members of The National Foundation for Eye Research (NFER), which aims to promote cataract research and develop non-surgical interventions for cataracts. Both researchers have devoted decades of work to explore bottlenecks in antioxidant and ARI development for the treatment of cataracts. Equally important to the development of drugs is the clinical evaluation of their efficacy. This has been conducted primarily through subjective observations of improved visual acuity or lens changes observed by slit lamp. Dr Manuel Datiles in collaboration with Dr Rafat Ansari of NASA has developed and validated the use of the dynamic light scattering (DLS) technique to safely and non-invasively assess protein aggregation in the lens of human patients in vivo. This allows for the progressive quantitative measurement of cataractous lens protein changes which lead to increased light scattering within the lens even before such changes can be detected by standard ophthalmic instrumentation. Use of this technique could become the gold standard for human clinical trials for establishing the efficacy of anti-cataract agents.
Dr Zigler and his colleagues have studied the effects of oxidation on the lens over many years and identified several antioxidants including the patented clinical candidate Tempol H. Dr Kador has developed topical ARI Kinostat® and a number of synthetic multifunctional redox modulators (MFRMs) that reduced oxidative damage in the lenses of animals through multiple mechanisms. Since these will require years of additional preclinical and clinical studies before becoming available for humans, a nutraceutical formulation was further developed that is designed to mimic the chemical properties of the MFRMs by scavenging free radicals and selectively binding redox metals. This topical formulation containing four different nutraceutical antioxidants with cetrimide, a preservative to enhance corneal permeation, is currently available for the veterinary market as Optixcare® Eye Health. In animals studies this strong antioxidant formulation delays the development of cataracts, and also maintains normal tear flow in the development of dry eye disease. Clinically its strong antioxidant effects have been documented in the treatment of tear stains which are dark brown or reddish marks that appear beneath select dog’s eyes that are caused by the oxidation of iron present in their tears. Clinical observations by Veterinary Ophthalmologists also suggest that the formulation may be beneficial for delaying age related cataracts. (See therapeuticvision.com for more information). This suggests that the future development for humans, especially in underdeveloped countries, is possible after clinical validation with the DLS technique.
Personal Response
What are the plans for the application of Optixcare Eye Health and Kinostat® in humans?
To find out more about the National Foundation for Eye Research see https://www.cataractresearch.org/. Information about Kinostat and Optixcare Eye Health can be found at https://therapeuticvision.com.