Preventing nickel allergy using various topcoats for decorative items
If you have a nickel allergy, something as simple as wearing a watch can trigger an allergic reaction. Nickel can be found in everyday decorative items, including costume jewellery and watches, which can make it difficult for those with an allergy to avoid. A collaboration between Dr William Y. Lo and Clive Whittington aims to improve the use of nickel in the marketplace and help prevent nickel allergy.
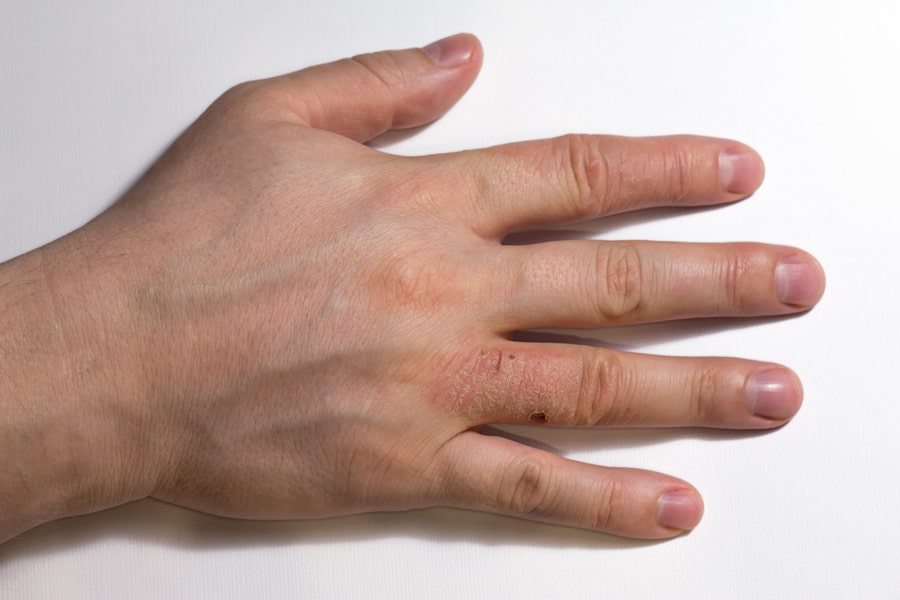
When a part of the body is consistently in contact with nickel, sweat can liberate nickel ions which are absorbed by the skin. That area of skin can become easily irritated and prone to an allergic reaction. This phenomenon is called Allergic Contact Dermatitis (ACD), but it is also colloquially referred to as nickel allergy. The skin turns red and itchy as a response to the allergen (nickel). Allergic sensitisation, leading to ACD, can often be triggered when the skin is in contact with nickel-plated watches, items such as clothing hooks, clasps and zippers, costume jewellery, and body piercing items.

Image credit: Kalcutta/Shutterstock.com
What causes ACD?
It’s difficult to tell who will become allergic to nickel or what can trigger contact dermatitis. In the UK, contact dermatitis affects 9% of the population and can be caused by many irritants. Nickel is a common allergen, but nickel allergy mostly affects females, in particular young women – often as a result of wearing more jewellery.
ACD does not occur upon first contact between the skin and nickel (the allergen). Instead, the body becomes sensitised, but may not react immediately. When the skin is further exposed to nickel, the now-sensitised body treats it as an allergen and the immune system reacts. The response to the nickel causes the skin to become red and itchy. Once the item containing nickel is removed, ACD symptoms will start to dissipate. The degrees of allergic response differ greatly from person to person.
Sweat from the skin can liberate nickel ions which are absorbed by the skin and initiate an allergic response
Clive Whittington and Dr William Lo evaluated non-nickel surface coatings which enable nickel-containing consumer products to be safely placed on the market without concern for nickel release. Such products must comply with specific regulations covered by the EU ‘Nickel Restrictions’ which impose limits on the amount of nickel released from articles intended to come into direct and prolonged contact with skin (like the back of a watch).

In the tests run by Whittington and Lo, each coating was measured for nickel release according to the CEN (Comité Européen de Normalisation, European Committee for Standardization) nickel tests for coated items: EN 12472 (for wear and corrosion) and EN 1811 (for the determination of nickel release). The CEN tests require that nickel release should not exceed 0.5 μg cm−2 week-1 after two years of normal use. However, because of measurement uncertainty an article is compliant if nickel release is < 0.88 μg cm−2 week-1 after two years and can be placed on the market.Since costume jewellery is usually made from cheaper metals and plated with nickel, these items are more likely to trigger nickel ACD in sensitised persons. Therefore, there is a need for efficient topcoats to prevent nickel release from these decorative items in particular.
Coatings for nickel-based decorative items
 In their ongoing research, Whittington and Lo look at using different coatings that prevent the release of nickel from nickel-based decorative items on the market. They tested non-nickel surface coatings including gold alloys, coloured and uncoloured topcoats against nickel release and found most coatings succeeded in preventing nickel release well within the CEN nickel-release limits.
In their ongoing research, Whittington and Lo look at using different coatings that prevent the release of nickel from nickel-based decorative items on the market. They tested non-nickel surface coatings including gold alloys, coloured and uncoloured topcoats against nickel release and found most coatings succeeded in preventing nickel release well within the CEN nickel-release limits.
Chromium topcoats
In their initial tests, the authors found that regular chromium deposited from a hexavalent electrolyte, microporous trivalent chromium from a chloride electrolyte, and ultraviolet (UV) cured polyurethane electrophoretic coatings prevent nickel release when applied according to the relevant International Organization for Standardization (ISO) specifications. However, hexavalent chromium electrolytes are constrained in many jurisdictions due to environmental concerns. Trivalent chromium electrolytes therefore present an effective alternative.

Sulphur-containing bright nickel with flash gold deposits
Whittington and Lo also tested electrodeposits of sulphur-containing bright nickel on pieces of costume jewellery and determined that nickel ions were released at a rate exceeding acceptable CEN limits. A thin, porous flash topcoat of gold applied over the same items also failed the tests, since the thin coating of gold significantly accelerated the release of nickel to figures exceeding the CEN limits. On the other hand, an uncoated gold alloy containing 6% nickel passed the CEN nickel-release tests.
The EU ‘Nickel Restrictions’ impose
limits on the amount of nickel released from articles.
Metal surface coatings
Costume jewellery and watch cases having an electrodeposited palladium barrier coating (either as a topcoat or an intermediate layer over bright nickel) prevented nickel release and kept it within CEN limits successfully.
Nickel-containing austenitic stainless-steel watch cases
For this test, Whittington and Lo utilised watch cases made of nickel-containing stainless steel (Grades 304 and 316). They masked off the areas of the watch case that did not come into contact with skin and tested for nickel release when uncoated and also coated with a very thin Physical Vapour Deposition Titanium Nitride (PVD TiN) coating. Both the uncoated and coated watch cases passed the CEN nickel release tests comfortably.
What’s a PVD TiN coating and how is it applied?
PVD TiN refers to a Titanium Nitride (TiN) coating applied by a process called Physical Vapour Deposition (PVD). This coating is the most commonly applied PVD coating because it is durable, inert, adherent and can be easily stripped and recoated if needed. A PVD TiN coating has several uses and is currently used on many expensive tools, medical (implanting and surgical) instruments, food processing and decorative applications. TiN is applied by vaporising the material from a solid source in the form of atoms/molecules and then transporting it in the form of vapour through a vacuum, low-pressure gas or plasma to the substrate (e.g. a watch case) where it condenses and coats the item.
Coatings can be coloured too…
As well as looking at the different coatings that prevent nickel release, Whittington and Lo also tested different colours that could be applied to topcoats to give the effect needed. For example, organic non-nickel topcoats may be dyed to produce a variety of colours in the surface finish of the item. The results from these tests showed that most coatings passed the CEN tests and did not exceed the nickel release limits.

Nickel is one of the most common metals used in costume jewellery and watch cases. As nickel allergy is so widespread, research to discover which coatings could be used to prevent nickel release is extremely significant.
The researchers found regular hexavalent chromium, microporous trivalent chromium from a chloride electrolyte, ultraviolet (UV) cured polyurethane electrophoretic coatings and an electrodeposited palladium barrier all prevent nickel release. Additionally, nickel-containing stainless steel with or without a PVD TiN coating readily passed the CEN nickel release tests. The results of this work mean these coatings and alloys can be used for decorative articles that are sold in the market without concerns about nickel release exceeding the recommended limits.

Personal Response
What first sparked your interest in nickel allergy and the prevention of ACD?