Tonic thermoelectric materials – powering future technology
Are you wearing a smartwatch? If so, what powers it? Chances are, it’s charged using mains electricity. But soon your body’s heat could be powering your wearable devices, using thermopower. Thermoelectric (TE) generators convert thermal energy into electricity. However, conventional TE generators typically can’t provide the voltage levels needed in small, wearable devices. But ionic thermoelectric materials (i-TE materials) can. The environment in which ions diffuse (the electrolyte) strongly influences electric output. The work of Professor Xiao-ming Tao at The Hong Kong Polytechnic University and colleagues including Dr Liu Jin on ion diffusion could help uncover new materials to power future technology.
One of the main bottlenecks in the production of personal electronic devices, including wearable electronics, sensors, and nodes for the rapidly growing Internet of Things, is the need for efficient, lightweight, reliable, and environmentally sustainable power sources. The Internet of Things refers to objects with inbuilt digital technology, such as sensors and software, which can connect with other systems and devices via the internet. One example is the Fitbit – a wearable bracelet that monitors users’ fitness activities and transmits data to an app that monitors performance against set goals.
As a result of increasing demand for devices like this, wireless or battery-free power supplies are gaining increasing attention as potential means of powering personal electronics, as they avoid the requirement of integrating traditional rechargeable batteries. In this case, electric power is generated by exploiting kinetic or thermal energy already present in the environment; for instance, sunlight, human motion, or environmental heat.

Thermoelectric generators are particularly promising in this context, as they can convert waste heat from the human body or the environment into usable electrical power. In principle, they can also be easily miniaturised (since they do not require moving parts). To help progress this emerging field, recent research by Professor Xiao-ming Tao at Hong Kong Polytechnic University and colleagues including Dr Lin Shuping, Dr Zeng Wei, and Dr Liu Jin has revealed robust theoretical models of ion diffusion, and could lay the foundation for creating new ionic thermoelectric materials – or with improved properties for energy harvesting.
The Seebeck effect
The thermoelectric effect is the direct conversion of a temperature gradient into an electric voltage (or vice versa). A thermoelectric generator creates a voltage when there’s a difference in temperature across it. In an electrically conductive thermoelectric material, an electromotive force, which induces the migration of electrical charge and subsequently generates net voltage, develops whenever there is a temperature difference between two points of the material. This phenomenon is known as the Seebeck effect, and the ratio between the electromotive force and the temperature gradient is known as the Seebeck coefficient.
It is possible to accurately describe the thermoelectric effect performance of materials. The conversion efficiency of a thermoelectric generator can be quantified using a dimensionless figure of merit (ZT), which is proportional to the square of the Seebeck coefficient. Materials with large Seebeck coefficients are the most desirable for the fabrication of thermoelectric generators.
Electronic thermoelectric generators
Conventional thermoelectric generators mainly use narrow-gap semiconducting materials, including compounds like Bi2Te3, SnTe, and SnSe, or metallic crystals like Cu2Se to create voltage differences. In these systems, the charge carriers responsible for the power generation are either electrons or holes, which migrate under the effect of the temperature-induced electromotive force.
These compounds are the most extensively studied class of thermoelectric materials. Usually, they exhibit very small Seebeck coefficients, meaning that – at room temperature – the voltage they produce remains extremely low. Therefore, their use in conventional electronic devices is currently impractical. An unfeasibly huge number of thermoelectric cells or a voltage booster would be required to achieve the voltages (typically of the order of several volts) needed for powering devices.
Controlling and optimising the thermoelectric performance of electronic thermoelectric materials is also challenging, owing to the complex interplay of the Seebeck effect with electrical and thermal conductivity. Additionally, these materials are typically rigid in their bulk forms. They can’t easily be integrated into devices as flexible structures (like thin films and fabrics), which makes them unsuitable for wearable device fabrication.

Ionic conduction
With this in mind, Tao and her colleagues aimed to address some of the limitations of electronic thermoelectric materials – including their low electric output and rigidity – by considering a class of systems known as ionic thermoelectric materials. In these materials, an electric field is generated by the migration of anions or cations (negatively or positively charged atoms or small groups of atoms) instead of holes or electrons. These ions diffuse within a fluid medium, typically a liquid solution or a gel containing the ions with opposite charges, known as an electrolyte. Tao explains, ‘To overcome restrictions of electronic thermoelectric materials, we have explored other types of materials, including organic flexible TE materials which also use electrons or holes as charge carriers. We have also focused our research on discovering new ionic thermoelectric (i-TE) materials by simply changing the charge carriers to ions.’
Power generation in ionic thermoelectrics
Two very different mechanisms explain the creation of a net voltage difference in iTE materials, or generators. The first is called the thermodiffusion effect (or Soret effect), which describes the diffusion of ions under a temperature gradient. The second mechanism is known as the thermogalvanic effect. It is caused by specific chemical reactions (redox reactions) involving a transfer of electrons between ionic or atomic species within the electrolyte, which occur at different temperatures. Thermodiffusion should always be prioritised when considering these two diffusion mechanisms: it represents the most important source of generated output voltage. Tao explains, ‘Whichever effect the i-TE systems are mostly based on, the thermodiffusion effect of the ions cannot be neglected. This is one of the most important components of the generated output voltage.’
Improving thermoelectric efficiency
Previous research on iTE materials has heavily focused on the choice of ions, either to increase the thermal mobility of the different ions in the i-TE systems (to generate a higher thermopower), or by exploring the ion chemical effects (to improve the TE performances of the i-TE systems). However, Tao and colleagues show that the environment of ion transportation – the electrolyte – also plays an important role.
Thermoelectric generators can convert waste heat from the human body or the environment into usable electrical power.ITE materials represent a substantial step forward in energy harvesting from environmental heat. ‘These materials,’ says Tao, ‘normally possess a rather high thermopower (or ionic Seebeck coefficient), and their output voltage is typically much larger than conventional electronic thermoelectric materials.’ For example, a power output two orders of magnitude higher than conventional thermoelectrics has been achieved using a quasi-solid-state device using NaOH (which splits, or dissociates, into Na+ cations and OH– anions and are responsible for power generation) is dissolved in a polyethylene oxide solution confined in nanocellulose channels.
Despite these successes, the major limitation of iTE generators is the generation of continuous electrical power, especially an applicable continuous electric current. Whichever mechanism the iTE generator uses, the output current will drop after working for a certain time period, which limits the applications as power generator.

Tao found that for iTEs, higher figures of merit can be obtained using ionic gels composed of ionic liquids (salts in a liquid state, entirely composed of anions and cations, at temperatures lower than 100oC) and fluorinated polymers. In these systems, the larger thermoelectric power output is due to the accumulation of ions at opposite ends of a device; the output varies depending on the concentration gradient between the oppositely charged ions at the cold and hot ends.
Understanding ion transport
Ion diffusion in iTE materials is influenced not only by the chemical nature of the ions but also – crucially – by the presence of the electrolyte medium in which the anions and cations move. Electrolytes control the electronic, magnetic, and optical properties of a material. In addition, they can undergo phase transitions. For instance, they can change from liquid solutions to more viscous gels (figure 2a), which in turn significantly affects ion transport and, ultimately, the power output of a thermoelectric generator.
Giant thermoelectric enhancement
To gain insight into the role of an electrolyte in promoting ion diffusion, Tao and collaborators have analysed in detail the effects of phase transitions between liquid solutions and gels induced by both thermal effects and irradiation with UV light in aqueous LiCl-based materials supported by polymers capable of undergoing controlled phase transitions. They observed significant increases in both thermopowers (6.5-fold) and ionic figures of merit (23-fold) in phase transitions from liquid solutions to the gel, as well as a large drop in thermopower after the opposite change from gel to liquid, depending on the nature of the electrolyte. UV irradiation has also been shown to have a massive influence on the thermopower, and is attributed to a change in the microscopic structure of the polymer electrolyte through the formation of cross-links between polymer chains.
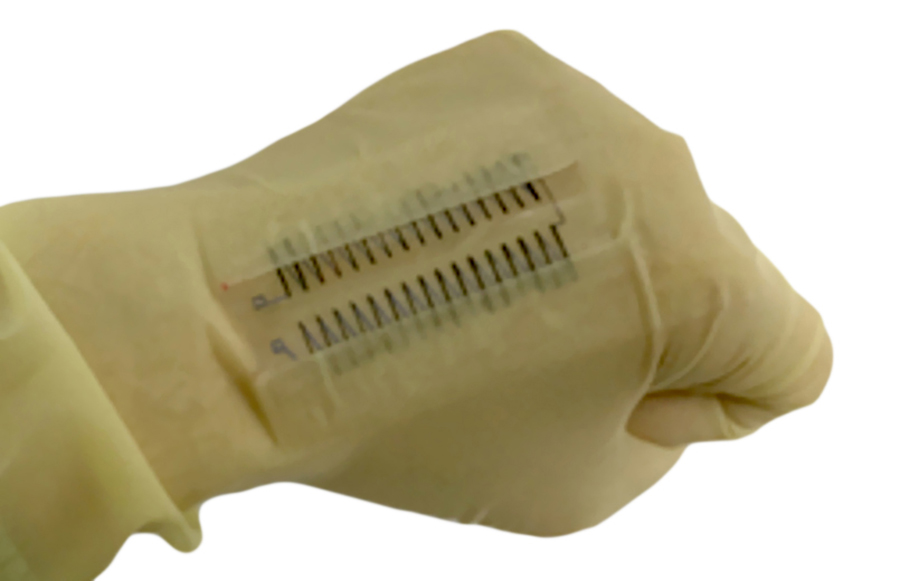
The role of phase transitions
In addition to demonstrating the massive influence of electrolyte phase transitions on the thermopower produced by iTE generators, Tao has developed a theoretical model describing the factors affecting ion transport. This can be used to account semi-quantitatively for all stages involved in phase transitions between solution and gel forms. The model enables the researchers to estimate the change in output thermopower during a phase transition using six parameters. These parameters can be independently probed and modified. Importantly, this allows for the control of output power and the identification of physical conditions leading to optimal thermoelectric performance.
A universal phenomenon
Even more importantly, the giant thermopower enhancement observed in Tao’s work demonstrates, for the first time, the existence of a universal dependence of ionic transport on phase transitions in polymer electrolytes. This finding opens new routes for fabricating thermoelectric generators – with performances far superior to traditional devices. ‘Our study,’ explains Tao, ‘sheds light on a novel and feasible way to improve energy conversion performance in ionic thermoelectric materials by controlled phase transitions – and may lead to new avenues in developing tuneable devices for low-heat energy harvesting applications.’ As a result, we may find ourselves wearing a fitness tracker in the future which was based on the very principles uncovered by this research.
Personal Response
How will your discovery of a direct link between a phase transition in an electrolyte and its thermoelectric performance shape the future of thermoelectric generator development and fabrication?Previous work has mostly focused on the species and chemical nature of ions, narrowing the research in this area. One of the most important findings of our work is that we demonstrated another aspect that can significantly affect the TE performances of iTE materials, apart from ion species. This universal phenomenon can provide a completely novel method to develop iTE generators and could even improve the TE performances of existing generators through the phase-transition mechanism.