Aromaticity in anions in fluorine-free ionic liquids
ILs composed of bis(trifluoromethyl sulfonyl)imide (TFSI) anion with different nitrogen-based aromatic and aliphatic cations – such as imidazolium, pyridinium, pyrrolidinium, piperidinium and quaternary ammonium derivatives – are widely tested in energy storage research. The quaternary ammonium cations and their derivatives, coupled with the TFSI anion, have good electrochemical stability at room temperature, but are thermally less stable than their quaternary phosphonium counterparts, and they would not be good electrolytes at high temperatures for LIB-SC systems.
Low viscosity is a primary requisite for electrochemistry since viscosity pointedly affects the ionic conductivity, diffusivity, dispersion, mixing, filtration, and even equipment selections. Despite the importance of lower viscosity and higher ionic conductivity, there have been limited attempts at lowering liquid viscosities of phosphonium ILs.
In this regard, we have designed tetra(n-butyl)ammonium and tetra(n-butyl)phosphonium-based ILs containing relatively small heterocyclic anions in an attempt to achieve the following desirable properties:
- Enhanced thermal stability: phosphonium-based ILs are known for their high chemical and thermal stabilities and comparatively low toxicity compared to their ammonium analogues.
- Faster diffusivity of ions: the investigation of ion self-diffusions is of great importance for ILs as electrolytes for electrochemical devices.
- Higher electrochemical stability: alkyl phosphonium-based ILs have wide electrochemical windows and higher ionic conductivities and in some cases better electrochemical properties than their alkyl ammonium counterparts.
The idea of aromatic anions functionalised with electron delocalised carboxylic functional group is explored to prepare halogen-free and highly stable ionic liquids as potential future electrolytes to be used in electrochemical devices. The synthesis of the thermally and electrochemically stable ILs is the primary motivation of our study, in which we focused on small phosphonium and ammonium cations and fluorine-free aromatic heterocyclic anions such as furoate [FuA] − and thiophenecarboxylate [TpA] − that can potentially be used as future electrolytes for electrochemical applications. Both the experimental and theoretical studies have been performed in order to understand the effect of functional aromatic anions on the physical and electrochemical properties of these ILs.
Abstract
Ionic liquids (ILs) composed of tetra(n-butyl)phosphonium [P4444]+ and tetra(n-butyl)ammonium [N4444]+ cations paired with 2-furoate [FuA]-, tetrahydo-2-furoate [HFuA]- and thiophenecarboxylate [TpA]- anions are prepared to investigate the effects of electron delocalisation in anions and the mutual interactions between cations and anions in their physical and electrochemical properties.
The ILs are arranged in decreasing order of their ionic conductivities as [P4444][HFuA] (0.069 mS cm−1) > [P4444][FuA] (0.032 mS cm−1) > [P4444][TpA] (0.028 mS cm−1) at 20 °C. The oxidative limit of the ILs followed the sequence of [FuA]- > [TpA]- > [HFuA]-, as measured by linear sweep voltammetry (LSV). This order can be attributed to the electron delocalisation in [FuA]- and in [TpA]- aromatic anions, which has enhanced the oxidative limit potentials and the overall electrochemical stabilities.
Findings
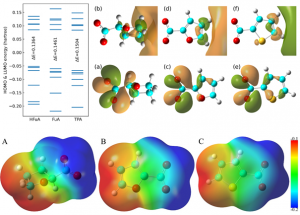
The synthesised ILs were characterized by 1H, 13C and 31P NMR and FTIR to confirm the structures and purity of the products, and also the aromatic characters of the anions. The charge delocalisation effect on these anions are further manifested on the HOMO (highest occupied molecular orbital) and LUMO (lowest unoccupied molecular orbital) energies. Figure 1 presents the molecular orbital energies from HOMO-5 to LUMO+5, the HOMO-LUMO energy gap, and the charge density contours of three anions at HOMO and LUMO states. The charge delocalization effect for three anions are characterized by [HFuA]- < [FuA]- < [TpA]-, and therefore the HOMO energies are gradually lowered from [HFuA]- to [TpA]- with a concomitant increase in LUMO energies and the enlarged energy gaps (ELUMO-EHOMO), indicating that the former is relatively easier and the latter is harder to oxidize.
This is further confirmed by the thermo-electrochemical stabilities of ILs consisting of these anions coupled with [P4444]+ cations. The variations of HOMO and LUMO energies are detailed in the corresponding electron density contours in Figure 1. The NMR, FTIR and the DFT calculations confirmed the electrons delocalization in [FuA]- and [TpA]- anions over the aromatic rings that might have a pronounced impact on the physiochemical and electrochemical properties of the synthesized ILs.
The development of thermally and electrochemically stable fluorine-free, cost effective, environmentally benign, and sustainable electrolytes will partially meet the challenges associated with safety, recyclability, accessibility, affordability and service life of LIB-SC technology.
References
1. Zhang J., Bond A. M., (2005), Practical Considerations Associated With Voltammetric Studies in Room Temperature Ionic Liquids, Analyst, 130, 1132–11472. Matsumoto H., Sakaebe H., Tatsumi K., Kikuta M., Ishiko E., Kono, M, (2006) Fast Cycling of Li/LiCoO2 Cell with Low-Viscosity Ionic Liquids Based On Bis(fluorosulfonyl)imide [FSI]−, Journal of Power Sources, 160, 1308–1313
3. Tsunashima K., Sugiya M., (2007) Physical and Electrochemical Properties of Room Temperature Ionic Liquids Based on Quaternary Phosphonium Cations, Electrochemistry, 75, 734–726
4. Fraser K. J., MacFarlane D. R., (2009) Phosphonium-Based Ionic Liquids: An Overview. Australian Journal of Chemistry, 62, 309–321
5. Shah F. U., Gnezdilov O. I., Filippov A., (2017) Ion Dynamics in Halogen-Free Phosphonium Bis(Salicylato)Borate Ionic Liquid Electrolytes for Lithium-Ion Batteries. Physical Chemistry and Chemical Physics, 19, 16721–16730
6. Matsumiya M., Suda S., Tsunashima K., Sugiya M., Kishioka S., Matsuura H., (2008) Electrochemical Behavior of Multivalent Complexes in Room Temperature Ionic Liquids Based On Quaternary Phosphonium Cations. Journal of Electroanalytical Chemistry, 622, 129–135
Written By
Inayat Ali Khan
Luleå University of Technology, Sweden
Contact Details
Email: inayat.khan@ltu.se
Telephone: ++46761409067