Preterm birth and danger-associated molecular patterns
Preterm birth is the leading cause of infantile mortality. Dr Claire Kendal-Wright from the Chaminade University of Honolulu works on human extra-embryonic membranes. More specifically, she studies danger-associated molecular patterns, or DAMPs, and examines how these molecules contribute to the prelabour rupture of the membranes that leads to birth. Understanding the mechanisms involved in healthy pregnancy could eventually allow the development of therapies aiming at preventing preterm birth.
Every year, worldwide, an estimated 15 million babies are born preterm; that is more than 1 in 10 babies. While a normal pregnancy lasts about 40 weeks, birth is considered to be preterm or premature when it occurs before 37 weeks of pregnancy are completed. The earlier a baby is born, the higher the risk of death or serious disability. Preterm birth is one of the most common negative pregnancy outcomes as the resulting complications are the leading cause of death among children under 5 years of age.
Rates of preterm birth vary across countries and range from 5% to 18% of babies born. The incidence of preterm birth differs even within countries: in the United States, much of the impact of preterm birth is borne by ethnic minority groups. Dr Claire Kendal-Wright, who works at the Chaminade University of Honolulu, particularly focuses her research on prematurity in Hawai’i and the Pacific Islands, where preterm birth is an issue mostly for the native peoples. Indeed, the infant mortality rate is twice as high for Native Hawaiian mothers compared to Caucasians, and almost half of this infant mortality is related to preterm birth.

Physiological changes leading to birth
As pregnancy comes to term, different physiological changes lead to birth: the cervix softens and dilates to allow the passage of the foetus, the myometrium (muscular layer of the uterine wall) contracts, the placenta separates from the uterus, and the rupture of the foetal membranes leads to what women commonly describe as “waters breaking”. These distinct physiological events need to occur in a coordinated manner for the successful delivery of the foetus at term. Indeed, desynchrony or dysregulation of these events can lead to preterm birth.
Preterm birth can result from different events. Approximately 20% of all preterm deliveries are by Caesarean section for maternal or foetal indications. Of the remaining cases, around 30-35% are caused by premature prelabour rupture of the membranes (pPROM), 20-25% result from intra-amniotic infection, and the remainder are due to premature uterine contractions.
There is more and more evidence suggesting that cellular stress is the trigger for both foetal membrane weakening and labour.
The trigger that initiates the physiological changes leading to birth remains elusive. In approximately 60% of preterm births, the reason why delivery occurred earlier than expected is unknown. Understanding what happens in normal pregnancy and how labour onset is triggered is important because it can help explain how the physiological mechanisms may deviate and lead to preterm birth.
Foetal membranes
The foetal membranes (the amnion and the chorion) surround the developing embryo and form an interface between the foetus and the mother. The amnion is the innermost layer. It is in contact with the amniotic fluid, the foetus and the umbilical cord. The chorion separates the amnion from the uterus.
Amnion and chorion maintain the conditions necessary for foetal health and therefore protect the foetus during pregnancy. As pregnancy advances to term, the foetal membranes undergo weakening, in preparation for their rupture. The first step in the weakening is the separation of amnion and chorion which, during pregnancy, are in contact. Then, as a result of both physical forces and biochemical changes, the membranes can break. Rupture of the membranes usually triggers labour and delivery; that is when we talk about prelabour rupture of the membranes (PROM). The timing of PROM is crucial for healthy pregnancy outcomes. If the rupture occurs early, it can result in premature PROM (pPROM) and preterm birth. Foetal membranes and pPROM are the main focus of Dr Kendal-Wright’s work.
The role of cellular stress
There is more and more evidence suggesting that cellular stress is the trigger for both foetal membrane weakening and labour. Cellular stress occurs when cells are exposed to damaging environmental conditions. In return, cellular protection mechanisms are set into motion. One of these mechanisms is inflammation, which has been shown to occur during labour.
Distension of the foetal membranes, which stretch as the foetus grows, is a mechanical stressor that could trigger membrane weakening and labour. Oxidative stress is another potential stressor: as cells of the amnion age, harmful molecules accumulate and damage the cells. Both distension of the foetal membranes and oxidative stress are known to stimulate inflammation.
From cellular stress to PROM
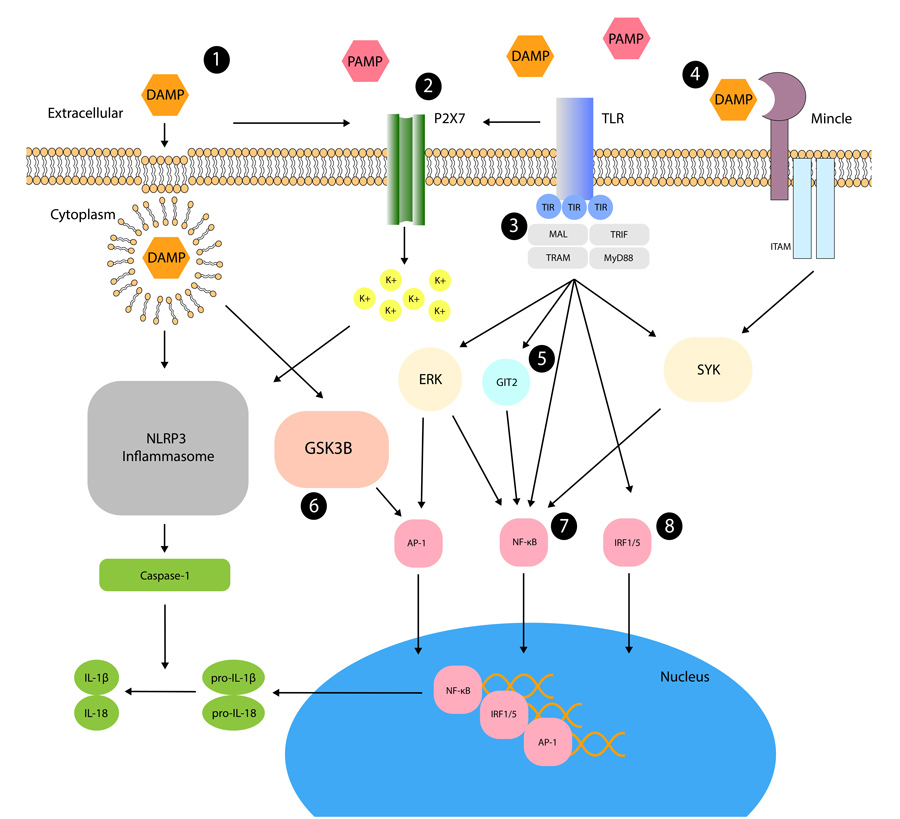
In response to tissue injury, damaged or dying cells can release or activate molecules named danger-associated molecular patterns (DAMPs). It is through DAMPs that distension and oxidative stress could stimulate inflammation and weaken the foetal membranes. Indeed, the general biological function of DAMPs is to raise the alarm within tissues, which they do by producing a number of proinflammatory cytokines.
The production of proinflammatory cytokines involves a cascade of biochemical reactions. Cellular stress first increases the production of DAMPs. Then, DAMPs interact with molecules named Toll-like receptors (TLRs), which activate the transcription factor NF-kB. As a transcription factor, NF-kB can modulate gene expression and stimulate the production of proinflammatory cytokines. By driving inflammation, proinflammatory cytokines promote cellular changes. Within the foetal membranes, components of the extracellular matrix are broken down, which physically weakens the membranes, making them more prone to rupture.
DAMPs in preterm birth
Cellular stress, DAMPs, proinflammatory cytokines, inflammation and rupture of the foetal membranes are involved in healthy pregnancy. However, while this cascade of events normally occurs after 40 weeks of pregnancy, it can be triggered prematurely, and pPROM can then cause preterm birth.
Within the foetal membranes, components of the extracellular matrix are broken down, which weakens the membranes and makes them more likely to rupture.
In preterm birth with pPROM, there is evidence that DAMP-driven inflammation is associated with foetal membrane weakening. Levels of DAMPs are higher in foetal membranes from women who had preterm labour or pPROM, and this can be due to different causes. Inflammation initiated by bacterial infection causes premature birth by preterm labour and pPROM. The initiation of this inflammation is mediated by pathogen-associated molecular patterns (PAMPs), which are very similar to DAMPs but originate from pathogens instead of damaged or dying cells. Like DAMPs, PAMPs interact with TLRs and promote inflammation. Likewise, viral infection elicits inflammation through the activation of TLRs.
While bacterial or viral infections can be involved, they are not necessary: inflammation in the absence of infection (called sterile inflammation), which likely occurs due to cellular stress, can also lead to pPROM. Different causes can thus increase levels of DAMPs, and higher levels are related to higher risks of preterm birth.
An example of DAMP
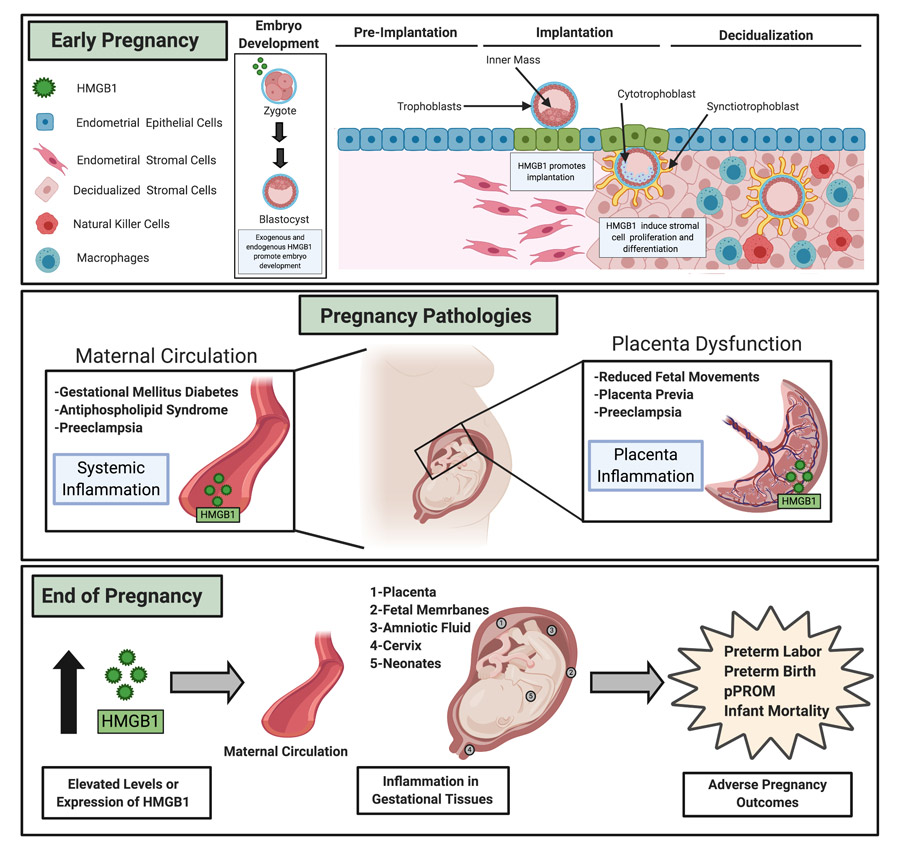
One DAMP, named high mobility group box 1, or HMGB1, has been particularly studied. Its levels are elevated in membrane extracts from preterm labour compared to term labour. HMGB1 can be released in a sterile environment or secreted in response to infection. It increases levels of proinflammatory cytokines and is likely to promote foetal membrane weakening.

HMGB1 has been shown to play a critical role not only for labour onset but also during the early stages of pregnancy. Indeed, a proinflammatory response driven by HMGB1 is essential for the development and implantation of the embryo. HMGB1 levels then need to be tightly regulated throughout pregnancy as high levels within the uterine cavity have been linked to pregnancy failure. In addition, chronic inflammation, resulting from increased HMGB1 within the maternal circulation and gestational tissues, also increases the risk for preterm birth or infant mortality.
A delicate balance
The inflammatory response mediated by DAMPs such as HMGB1 in the early stages is critical for pregnancy success, but excessive DAMPs levels within the uterine cavity and expression within the placenta have been shown to lead to pregnancy loss.
Once the embryo is implanted, an anti-inflammatory environment is key for the maintenance of pregnancy up until the initiation of labour. Thus, during the greater part of gestation, elevated DAMPs levels can promote the inappropriate proinflammatory signalling that is central to many pregnancy morbidities such as preeclampsia, gestational diabetes and placental inflammation.
Finally, at the end of pregnancy, inflammation is needed for the normal process of birth. But once again, the balance is delicate since excessively high levels of DAMPs could initiate delivery too early and lead to preterm birth or infant mortality.
DAMPs as therapeutic targets
Due to the link between DAMPs and several pregnancy-related pathologies, the possibility of use DAMPs such as HMGB1 as a biomarker has been assessed. However, further research is needed to examine whether HMGB1 inhibitors could reduce inflammation within pregnancy, so that preterm birth could be avoided.
Personal Response
Do you have any idea why the burden of preterm birth weighs so much on native peoples from Hawai’i and the Pacific Islands?
This is not understood, it is likely to be a combination of our unique mix of socioeconomic and health issues here in the islands.
What are you planning next for your research?
I aim to understand how DAMP signals from the growing foetus can communicate towards the mother to initiate inflammation and tissue weakening in the foetal membranes.