How treatment epidemiology can shape future healthcare research
Epidemiology can be defined as ‘the study of what comes upon the people’ (from the Greek epi-demos-logos) and is best known as the science underpinning public health. Historically, epidemiology has focused on the surveillance of disease, also referred to as ‘disease epidemiology’, studying what level of disease occurs and in which populations. Disease epidemiology identifies changes in disease over time, as well as risk factors for disease occurrence, and disease prevention efforts. As science and medicine have evolved, a natural extension of disease epidemiology is the study of how diseases are treated, a subject area which Dr Aisling Caffrey, University of Rhode Island, has coined ‘treatment epidemiology’. This field of study has a number of different names, including pharmacoepidemiology, health services research, real-world evidence, outcomes research, health outcomes research, or pharmaceutical/device health outcomes research. Most simply, this field studies the effects of healthcare interventions to treat disease, and all these other terms fall under the umbrella of ‘treatment epidemiology’.
Treatment epidemiology
In treatment epidemiology, researchers study what types of treatments are used, including medications, medical devices, and alternative therapies, as well as other health services and health-system interventions. Treatment epidemiology studies how these treatments are prescribed by providers; for example, whether they are prescribed according to clinical guidelines or based on indications approved by drug authorities, and how they are used by patients, such as whether they are being refilled on time. Treatment epidemiology also studies the benefits and harms of treatments. These studies investigate whether medications are working in real-world clinical practice as they were intended to work, by alleviating symptoms or improving clinical measurements, without causing significant adverse events.
In exposure mapping, all treatments for a specific disease are mapped out for each patient over a period of days or months, depending on the expected duration of treatment.
An important aspect of understanding how treatments work in real-world clinical practice is understanding how diseases are treated, and variations in the treatment of the same disease. Patients may wonder why they were prescribed a certain medication for their high cholesterol, whereas their family members or neighbours were prescribed something different for the same condition. We call these variations ‘treatment heterogeneity’. Medication utilisation in real-world clinical practice and the magnitude of treatment heterogeneity, including the way providers and patients arrive at treatment decisions, are poorly understood. There is an assumption that patients with a specific condition receive approximately the same treatment, particularly if clinical guidelines exist. However, Dr Caffrey’s recent work shows that in fact, even patients with the same disease receive different treatments. “Therefore, there is a need to better understand what treatment in real-world clinical practice actually looks like across diverse clinical settings and diverse patient populations, to inform the design of future research.”
Treatment heterogeneity and exposure mapping
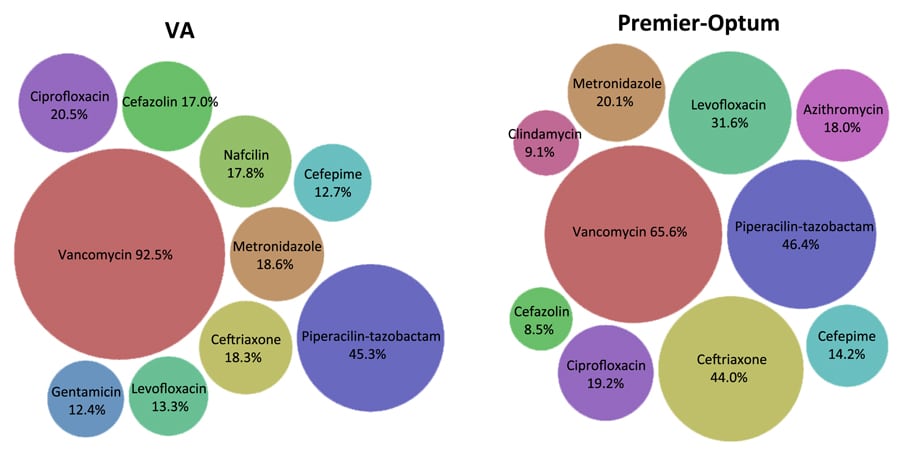
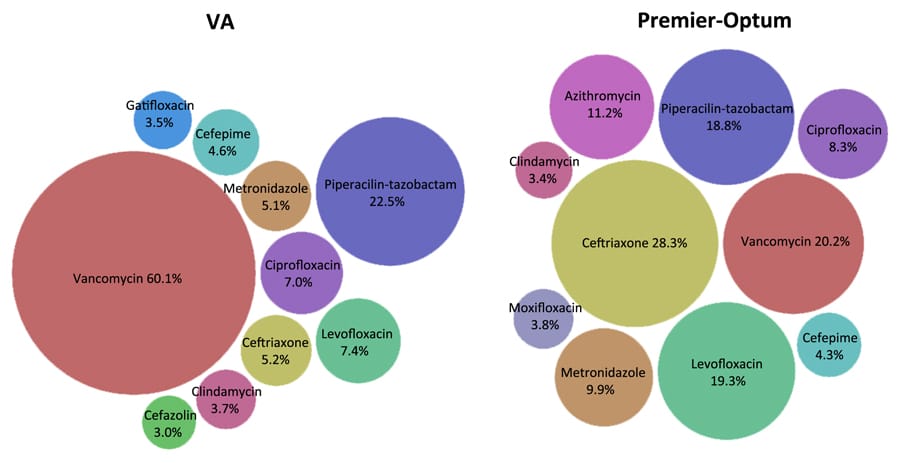
Dr Caffrey has developed methods that allow her to identify patterns of which medications are being used to treat specific diseases, called exposure mapping. “In exposure mapping, all treatments for a specific disease are mapped out for each patient over a period of days or months, depending on the expected duration of treatment.” This allows Dr Caffrey to describe common treatment patterns, including treatments used in combination and changes in treatments over time. Dr Caffrey has applied these methods to study antibiotic treatment patterns for serious bacterial infections. This is particularly important as antibiotic resistance continues to grow, and there is often a lack of evidence regarding the safest and most effective antibiotic regimens. Serious bacterial infections are complicated to treat, since the cause of the infection is unknown during the initial period of treatment and clinical symptoms change day-to-day, which leads to multiple changes in therapy every few days. This creates barriers to accurately defining antibiotic exposures in infectious diseases and linking specific treatments to clinical effectiveness.
Dr Caffrey aims to improve our understanding of treatment patterns using exposure mapping, and one of her recent projects described unique treatment patterns among patients with bloodstream infections. This study used a retrospective cohort study design to analyse hospitalisations at Veterans Affairs Medical Centres and community hospitals which had already occurred between 2002 and 2015. The Veterans Health Administration represents a US agency that provides healthcare services to military Veterans.
Over 50,000 hospitalisations with bloodstream infections, also known as bacteraemia, were included in the analysis. Dr Caffrey found that “for every 100 bacteraemia hospitalisations with changes in antibiotic therapy, 95 had unique antibiotic treatment patterns.” When considering hospitalisations with (88.5%) and without (11.5%) changes in therapy, overall, only 14% had the same treatment pattern as another hospitalisation. This study was the first of its kind, revealing a significant amount of treatment heterogeneity for bloodstream infections in real-world clinical practice, even for infections caused by the same organism (i.e. Staphylococcus aureus). Dr Caffrey has identified similar levels of extensive treatment heterogeneity in other infection types (e.g. pneumonia), and with other organisms (e.g. Pseudomonas aeruginosa). This work uncovers the inaccuracy of commonly used definitions of treatment in infectious diseases research.
Serious infection and continuation of medications
Other work done by Dr Caffrey has investigated the potential protective effects of statins, a medication normally used to lower cholesterol levels, in infectious diseases. Many of the existing studies show varying results and Dr Caffrey hypothesised that this was due to differences in exposure time to statins, and when treatment was started in relation to the onset of infection. Therefore, several of her projects have assessed the effect of statins on serious infections, investigating whether statin use was continued throughout periods of serious illness, and if so, what was the impact of this continued statin use.
For every 100 bacteraemia hospitalisations with changes in antibiotic therapy, 95 had unique antibiotic treatment patterns.
One of Dr Caffrey’s studies assessed the impact of continuing or discontinuing statin therapy during hospitalisations for bloodstream infections caused by a bacterium called Staphylococcus aureus. This study was conducted among Veterans Affairs patients and continuation of statin therapy was associated with significant beneficial effects on mortality. Dr Caffrey found that one death could be prevented in the first month after infection by continuing statin therapy in 10 patients during their hospitalisation.
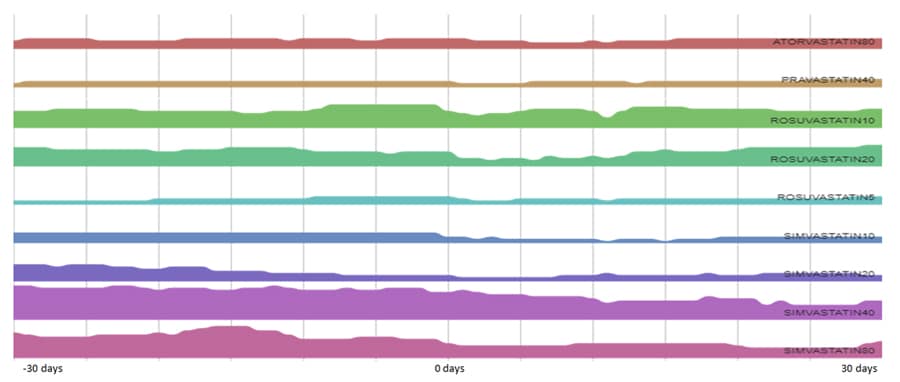
Dr Caffrey’s other exposure mapping work has shown that such hospitalisations for serious bacterial infections interrupt chronic disease medications, resulting in treatment heterogeneity. When evaluating the month before hospitalisation and the month thereafter among patients hospitalised with Staphylococcus aureus bacteraemia, only around one third of patients continued the same statin medication and dose for the 61-day observation period. A small proportion, around 7%, of patients had their statin drug or dose changed during the observation period. Statins were initiated in 10% of patients, and also discontinued in a similar proportion of patients. Overall, two thirds of patients experienced changes to their statin therapy, whether this was a discontinuation of the drug, a gap in therapy, or a change in the drug or dose used. This study provides an example of how exposure mapping could be used to improve operational definitions of exposure. In this case, how statin use changes before and after hospital admissions for a bacterial infection. Understanding more about these patterns of medication utilisation ultimately improves research in this field, allowing the identification of effective and safe healthcare interventions and enabling the discovery of better ways to treat diseases in the future.
Conclusions
Dr Caffrey concludes: “We have a limited understanding of how diseases are really treated in clinical practice, including which medications are commonly used, at which doses, and when changes in treatment are made.” When substantial treatment heterogeneity exists, meaning few patients receive the same treatment, it can be difficult to draw conclusions from studies which are not accurately defining treatment by using overly broad definitions. More specific treatment definitions, utilising exposure mapping and accounting for treatment heterogeneity, enable direct comparisons between patients but until these methods are implemented across the board, such studies should be interpreted with caution.
To improve upon this, Dr Caffrey has created the RX-OME ProjectTM, “to discover the nature of variability in drug prescriptions”. The RX-OME ProjectTM will use exposure mapping to identify patterns of usage for medications, including doses and duration of use. Learning more about the nature of treatment variability will help researchers design better studies and improve evidence to support prescribing practices and clinical guidelines.

Personal Response
Can you tell us more about the RX-OME ProjectTM?
To learn more about the RX-OME ProjectTM, please visit https://web.uri.edu/the-rx-ome-project/