Do bees farm microbes? Rethinking what it is to be a bee
Bees have not been eating what we thought they were. The work of Dr Prarthana Dharampal and Dr Shawn Steffan, University of Wisconsin, USA, shows bees are not strict vegetarians requiring just pollen and nectar for food. Larval bee health is also reliant on eating the microbes that are feeding on the pollen supplies. The bee larvae are further up the food chain than we thought, acting a lot more like livestock farmers than grazers. This is an exciting new perspective with implications for the use of fungicides on bee-pollinated crops.
Here we showcase the work of Dr Prarthana Dharampal and Dr Shawn Steffan at the Steffan Lab, University of Wisconsin-Madison, USA. Together with collaborators, they have set out to explain why seemingly ‘bee-safe’ agrichemicals were making bee larvae sick. This has led to a complete rethink on what it even is to be a bee!
Bees as mutualists
The give, take, bribery, and cajoling of food and energy exchange between living organisms has long been recognised as a complex web rather than a simple linear food chain. Bees were already known to be great collaborators within this system, managing relationships with flowering plants and occasionally humans. As children, we learn how flowering plants often make nectar and pollen to entice various types of bees to visit them and in return, bees move the pollen between flowers. This fertilisation service is often crucial for farmers to get decent crop yields.
In this narrative, the bees are vegetarians living on nectar and pollen coming straight from the plants, which are in turn fuelled by sunlight. We are the farmers growing the crops, occasionally maintaining beehives to promote pollination rates, and all is working smoothly. Well, it was. In the last 15–20 years, however, bee populations have been decreasing dramatically.
A newly discovered threat
Habitat loss, decreased genetic diversity, and the dramatic increase in the use of chemical pesticides (eg, neonicotinoids) have all been shown to impact not only the commercially managed honeybees and bumblebees, but also many native species of solitary bees in North America. Steffan and collaborators previously showed that bumblebee colonies that ate pollen contaminated with fungicides (agrochemicals used to kill fungal pests) produced fewer new worker bees and smaller queen bees. This was unexpected. Also, the kinds of microbes usually present within the bee colony had changed as a result of exposure to the fungicide.

Some of the routinely used fungicides had been safety-tested on the pollinators wherein adult bees were found to cope well when exposed to these products. Yet, bee populations that were exposed to such ‘bee-safe’ compounds were still dwindling. What could be the role of fungi in bee health?
Bees and their microbes
Adult bees generally get the bulk of their carbohydrates from foraged nectar and work in nutritional mutualism (both partners gain food and/or nutrients from each other) with their gut microbiome. Any effect the fungicides were having didn’t appear to be at this stage of bee–microbe symbiosis.
Could we consider the bees to be farmers in their own right, sowing a crop of microbes to sustain their larvae?Bee larvae, however, are mostly supplied with chewed-up pollen that has been softened with some nectar. It is common in both social and solitary species for this mixture to be left to age, and any microbes present, including fungi, will get a chance to multiply before a bee egg is laid on top. Since the hatching larvae lack the characteristic gut microbiome needed to digest raw pollen, maybe they need a healthy microbiome to be living inside the pollen provision to transform it into something more digestible?

Spying on developing bee larvae
Most ground-nesting bees dig little holes in the ground to use as their nurseries, but the blueberry bee (Osmia ribifloris) is a solitary bee that conveniently makes its brood cells in hollow reeds of phragmites grasses. Steffan and Dharampal flicked the eggs out with a paintbrush and reared them in sterile plastic wells lined with tiny tin cups full of pollen provisions. The use of these artificial brood cells made it possible to then observe and even remove and measure the larvae over time. The pollen provision was sterilised and then re-inoculated with varying amounts of live microbes. Treatments and controls were set up to test whether bee larvae needed the full complement of pollen-associated microbes within their diets to complete development.
Interestingly, the larvae that fed on provisions lacking microbes suffered heavily and did not grow properly. Despite having plenty of pollen available, fewer larvae made it to adulthood, and those that did were small and weak (slower growth rates, lower biomass, and decreased survivorship were recorded).
Dharampal and Steffan wondered if the microbes themselves made up a large part of the larval diet. Could we consider the bees to be farmers in their own right, sowing a crop of microbes to sustain their larvae?

How vegetarian are we really?
Luckily it is possible to establish how far removed an organism is from the original source of the sun’s energy (increasing trophic level) by measuring the profile of certain stable isotopes. Nitrogen isotopes are a useful trophic biomarker since the lighter 14N is more likely to be excreted during the organism’s life than its heavier counterpart, 15N. Animals living entirely on plant material are expected to have a smaller ratio of 15N to 14N incorporated into their amino acids (protein building blocks) than the carnivores eating those herbivores.
But what about an animal that is eating fungi? In a restaurant, we might happily label a stuffed mushroom as a vegetarian dish, but from a food-web perspective, the mushroom is a trophic level up from the leafy salad around it. If we consider the movement of the original energy from the sun, eating a mushroom is more similar to eating a hamster than eating some lettuce – since, like the hamster, the mushroom has itself been feeding on plant matter.
When Dharampal and Steffan performed stable isotope analysis on their bee larvae, they indeed found an isotopic composition that indicated that these larvae were not pure vegetarians and could be more accurately described as omnivores since they eat a significant quantity of microbes, which in turn were feeding on the pollen. Researchers had previously missed this critical food source, largely because it was invisible.
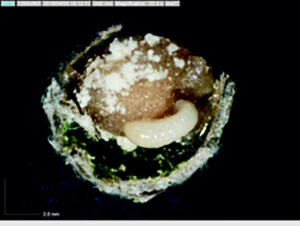
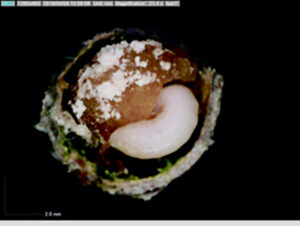
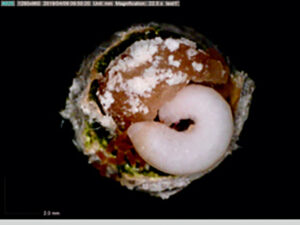
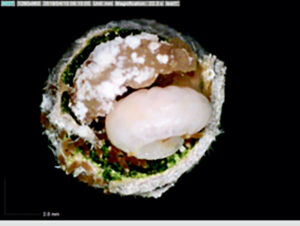
Bringing microscopic detritivores to the centre stage
Even before green plants had evolved, bacteria, protozoa, and fungi formed the first food webs. Detritivory (eating dead animal and plant matter) was a key part of the system and still represents the dominant trophic mode (source of nutrition) on Earth. Despite their large number and vital function, detritivores are not always given the detailed attention they deserve.
Like the bee example described above, the bulk of the organisms in so-called ‘brown ecosystems’ (organisms living on dead material) are microbes. However, there has been an inherent bias in our knowledge that favours the living, so-called ‘green food-web’, and the macro over the micro: think of how much easier it is to record a brightly coloured bird eating berries than it is to describe the diversity of microbes that breaks down the leaf litter under that tree.

Steffan and Dharampal are not put off by the considerable challenge of investigating the ultimate fate of the undead. By establishing who is really eating what, they are getting an increasingly accurate picture of the true flow of energy through ecosystems. Unravelling these unexpected relationships might reveal new selection pressures that drive the form, behaviour, and diversity of organisms.

How fussy are bees about their microbiomes?
Given that the bee microbiome appears to be such a key part of bee nutrition, Dharampal and Steffan decided to test whether a bee species might respond differently to microbiomes of their diets as allocated by adults of their own species compared to that provided by a different, but closely related type of solitary bee.
Dharampal and Steffan found that bee larvae were not pure vegetarians and could be more accurately described as omnivores.The bees were reared on pollen provision collected by either their own species (‘usual pollen’) or that collected by a different bee (‘unusual pollen’). The microbes were manipulated by killing the original microbiome, but then each pollen sample was then re-inoculated with either its original microbiome (‘usual microbes’) or the microbiome that had been foraged by the other species (‘unusual microbes’). When larvae were fed their usual pollen diet, as long as a microbiome is introduced, the type of microbes doesn’t seem to matter much. However, when fed the wrong pollen, having their usual microbiome appeared to be extremely critical for bee survival. The researchers conclude that for developing bees, the identity of microbial symbionts within pollen provisions is just as critical as the type of pollen for developmental success.

The work that Dharampal and Steffan have been doing is a wonderful demonstration of the practical benefits that can be gained from taking a much closer look at the microbial ecology of detritivores. Now that we have a clearer understanding of the flow of energy resources from sunlight to bees through microbial intermediaries, we can hopefully develop bee conservation strategies that would also safeguard their microbial symbionts. Changing the specific fungicides or even the timing of spraying might allow us to tread more lightly on these bee-friendly microbes and ensure we don’t inadvertently break up their crucial partnerships.
Personal Response
Does our increased understanding of the complexity of food webs and the role of microbes mean that the distinction between herbivore and carnivore is less meaningful than we thought?We know that as primary producers, plants have a trophic position of ~1.0. From a food-web perspective, however, living plant biomass is no different from non-living plant biomass. Whether alive or dead, the trophic position of a leaf is ~1.0. When said leaf is eaten by a grazer off the twig or decomposed by a microbe as it lies on the ground, both grazer and microbe will have an expected trophic position of 2.0, both are primary consumers (aka ‘herbivore’). Similarly, a wolf hunting elk is just as much of a carnivore as the microbes eating elk carcass. Therefore, the dichotomy between food that is alive (green food web) versus food that is dead (brown food web) becomes somewhat less meaningful; food is food, alive or dead, and for the most part, microbes can eat it all.
What are your next research goals? Will you be applying this approach with other taxa?
We are looking to delve deeper into the tripartite symbioses between angiosperms, bees, and microbes. More specifically, we are interested in figuring out how beneficial microbes continue to be exchanged between bees and flowers with such remarkable consistency and fidelity. Adopting an unorthodox view of bee pollination services, we ask:
• how much do bees forage for pollen compared to the microbes therein?
• what holds the microbial zoo inside a bee’s nest in check and keeps it from turning into a cesspool of pathogens?
• where do these beneficial bee-associated microbes overwinter and how are they reacquired each year anew?