Diagnosing Sjögren’s syndrome with salivary gland ultrasound
Sjögren’s syndrome is a chronic autoimmune disease, where the immune system gets overstimulated due to an unknown origin and attacks healthy parts of the body. In primary Sjögren’s syndrome (pSS), the immune system first attacks the glands that make tears and saliva, the lacrimal and salivary glands respectively, before manifesting in other organs. Therefore, pSS is characterised by lymphocytic infiltration, which is the build-up of white blood cells in the body. The usual symptoms are dry eyes and mouth with extra-glandular manifestations, such as fatigue and pain in the joints. In the UK, the syndrome usually manifests in patients aged 40-60 years old. Globally, it is present in 0.05% of the general population.
On the other hand, juvenile Sjögren’s syndrome (jSS) is uncommon and poorly defined compared to pSS in adults. Patients are usually diagnosed at 10 years but challenges remain to ensure the diagnosis of patients is accurate. Recurrent inflammation of the parotid gland (parotitis) is the general sign at the onset of jSS. This symptom and the presence of characteristic autoantibodies, such as anti-Ro/La (anti–Sjögren’s-syndrome-related antigen A autoantibodies) and RF (rheumatoid factor), are used as positive diagnostic tools in many patients. However, parotitis is a common disorder in children and can be of bacterial or viral infectious origin instead. Therefore, patients who present parotitis as a symptom can be mistakenly diagnosed as contracting an infection. To add complexity, there is later onset of some symptoms, which are dry eyes and mouth, in some younger patients and extra-glandular manifestations only occur in 50% of patients. Therefore, jSS is possibly an underdiagnosed disease, as both patients and doctors may not notice it until later in life.

Due to diverse clinical manifestations in younger patients, there is currently no available set criteria to diagnose jSS patients, and the American-European Consensus Group (AECG) or the American College of Rheumatology/European League Against Rheumatism (ACR-EULAR) are neither sensitive nor specific for jSS diagnosis.
Diagnosis of Sjögren’s syndrome
At present, there is no available cure for the disease. Many have hypothesised the causes of the syndrome, from genetics to hormone imbalance, but researchers are unable to pinpoint a specific cause. Treatments usually aim to manage the symptoms, including eye drops to keep eyes wet and saliva substitutes, such as sprays and lozenges. Methods of diagnosis and assessment include salivary flow measurement, minor salivary gland biopsy, sialography (radiographic examination of the salivary gland), scintigraphy (gamma scan), CT and/or MRI.
Salivary gland ultrasound is non-invasive, widely available, relatively cheap, and has a high resolution.
In patients with the absence of characteristic autoantibodies, biopsies are usually performed, which is invasive, with low sensitivity and variable interobserver reliability. Patients with dry eyes and mouth (sicca symptoms) are assessed using salivary gland scintigraphy, which utilises a radioisotope attached to a drug and the subsequent gamma radiation emitted is measured.

In summary, these tools are invasive and involve ionising radiation, which remains a challenge for patients living with pSS or jSS. Therefore, a bottleneck remains to find a safe alternative to diagnose patients.
Salivary gland ultrasound
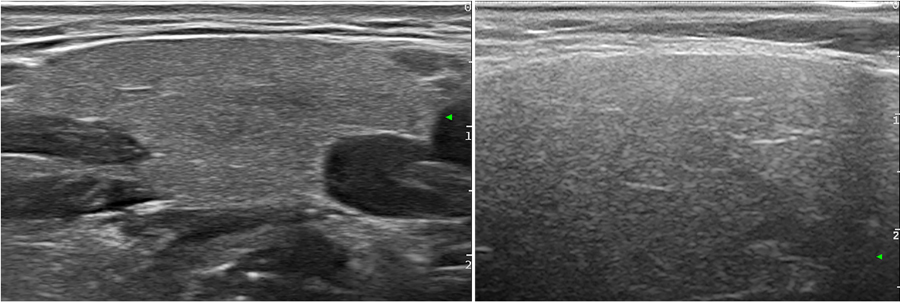
Salivary gland ultrasound (SGUS) evaluates the structure of the salivary glands by detecting changes in sound waves. It is an appealing alternative or can be used as a supplement to other methods to diagnose Sjögren’s syndrome. This is because it is non-invasive, widely available, relatively cheap, and has a high resolution. The procedure can be repeated multiple times to ensure accuracy of diagnosis and is available as an outpatient visit, which gives more flexibility for patients.
Dr Juan Carlos Nieto-González is a distinguished rheumatologist from the Hospital General Universitario Gregorio Marañón who has proposed the utilisation of SGUS in diagnosing and assessing pSS and jSS patients for many years. Dr Nieto-González has authored multiple papers to provide increasing support of SGUS to show the consistent echo-structural abnormalities of patients and its high sensitivity in classifying Sjögren’s syndrome.

For example, results from SGUS in multiple adult patients showed a consistent pattern, such as similar internal echotexture (structure of tissue layers during an ultrasound) and echogenicity (signal intensity in ultrasounds; image that appears brighter is more echogenic, which reflects higher sound waves). There was also the presence of many different sizes of oval dense tissue layers (called parenchymal inhomogeneity with multiple focal hypo-anechoic rounded areas) during the ultrasound scan. These characteristics using SGUS have been proposed by Dr Nieto-González to be included in the diagnostic criteria of pSS, as they are consistent across many patients. Similar results have been observed in children with jSS, which may make the diagnostic criteria using SGUS applicable to all age groups. However, there are also other diseases that show similar pathologies, such as amyloidosis and Ig4 syndrome.
Previous studies found that inclusion of SGUS information in the diagnosis of pSS to the AECG criteria increased sensitivity from 77.9% to 87% while inclusion to the ACR criteria increased sensitivity from 64.4% to 84.4%. These studies showed that SGUS may be a feasible and reliable diagnostic tool, which can be used alongside current diagnostic methods.
Salivary gland ultrasound may be a potential tool to reliably diagnose and assess primary and juvenile Sjögren’s syndrome patients.
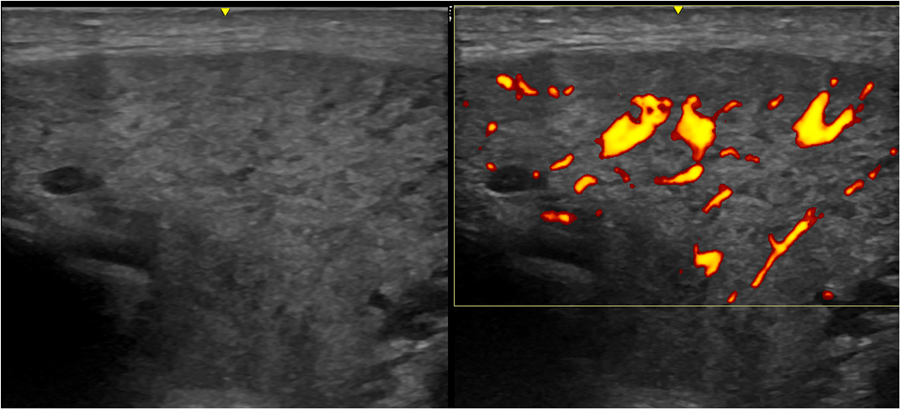
Dr Nieto-González and his colleagues aimed to assess multi-observer reliability of ultrasonographers with varying levels of working experience using SGUS on patients with established pSS. This clinical study was investigated at two European centres. In a 2016 paper published in The Journal of Rheumatology, they found that there was a high reliability in determining normal and/or abnormal salivary glands in pSS patients. There was also increased agreement in assessing parenchymal inhomegeneity of salivary glands between experienced ultrasonographers compared to inexperienced ultrasonographers. However, it takes relatively little time to assess patients using SGUS (approximately 5-10 minutes). Therefore, inexperienced ultrasonographers can easily be provided with extensive training for more accurate readings. However, there was significantly less reliability when assessing Doppler signals (measurement of blood velocity; increased blood flow and vascularisation are correlated with increased salivary glands inflammation in pSS). This study showed that SGUS is a reliable tool to assess the structure of salivary glands in pSS patients during follow-up visits but do not infer the reliability of SGUS in early diagnosis of pSS. More research is needed with larger patient groups, as these clinical studies have been conducted with a relatively small sample size.

As diagnosis criteria and symptoms for jSS have not been properly characterised in a large cohort of patients, Dr Nieto-González and other researchers conducted a clinical study, which was published in Arthritis Care & Research in 2020. SGUS was performed on 67 patients with jSS from the onset of disease until they were 18 years old. The patients were of European and American nationalities, which ensures diversity and increases the representativeness of the clinical study to other regions of the world. Approximately 63% of patients (n=42/67) fulfilled either the AECG or ACR-EULAR criteria that are widely used to diagnose adult patients. In comparison, 61% of patients (n=41/67) showed signs of pathology when using SGUS for diagnosis. Moreover, patients that showed Sjögren’s syndrome pathology were positively associated with characteristic autoantibodies compared to patients who did not show Sjögren’s-syndrome pathology. These findings showed that the presence of antibodies and SGUS can be used together to increase the diagnosis of jSS patients. It can also be included in AECG and ACR-EULAR, narrowing the gaps to having a set of criteria for the diagnosis of jSS.
In conclusion, these results showed that SGUS may be a potential tool to reliably diagnose and assess pSS and jSS patients. However, the usage of ultrasound to diagnose and assess other connective tissues diseases have to be further investigated. The strong association between pathological SGUS and autoantibody positivity may determine a subpopulation of patients with potentially severe Sjögren’s syndrome, which may help researchers better understand ways to manage the disease. All in all, these results by Dr Nieto-González provide support for the inclusion of SGUS in the classification criteria of Sjögren’s syndrome in the future.

Personal Response
What other areas of research are needed to further support the utilisation of SGUG in Sjögren’s syndrome diagnosis?