Neuropathological changes in Alzheimer’s disease linked to four key proteins
Alzheimer’s disease (AD) is characterised by progressive and irreversible loss of memory and thinking skills. Using sensitive cutting-edge techniques, Professor Rafael Luján and his collaborators at Castilla-La Mancha University, Spain, have found changes in the number, distribution, and density of four key proteins in AD. Notably, these proteins affect the ability of nerve cells to process and transmit signals. Not only does their work provide vital clues to the neuropathological alterations in AD, but it could also inspire new treatments.
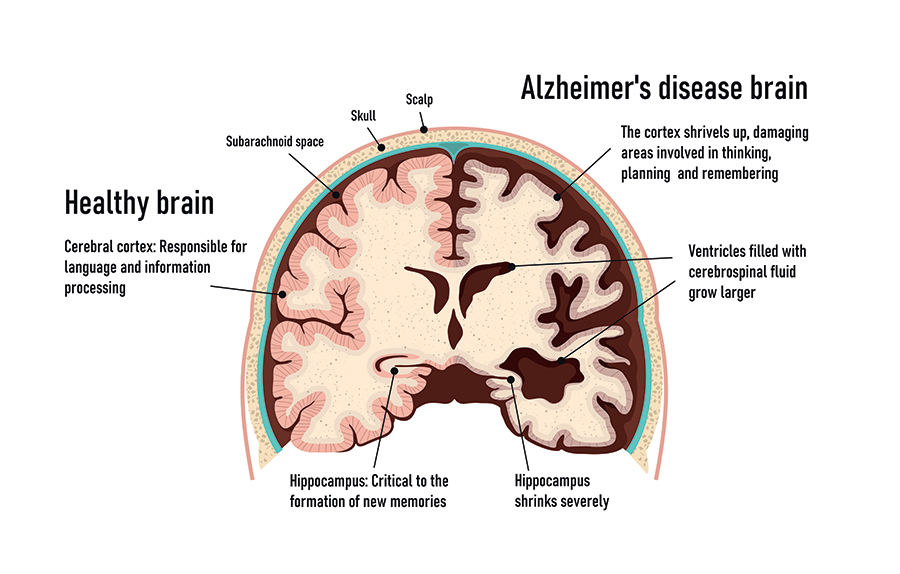
With the global population projected to continue ageing over the coming decades, Alzheimer’s disease (AD) is a serious and growing public health concern. Characterised by progressive and irreversible loss of memory and thinking skills, AD severely disrupts a person’s ability to function normally. The molecular mechanisms of AD are poorly understood, and few treatment options are available. In the brain, the defining features of AD are the build-up of a protein called β-amyloid (Aβ), and tangled clumps of tau protein. The strongest characteristic of AD, however, is a loss of synapses: the place where the messages between nerve cells and between nerve cells and our muscles take place.

The human brain contains an astounding 86 billion neurons (nerve cells). Neurons are the ‘information messengers’ of the brain and communicate with one another in a unique way. Through a process called neurotransmission, chemical messages pass between synapses (the small gap between cells). Synapses can be divided into two groups, excitatory and inhibitory. Excitatory synapses promote an action potential through the binding of glutamate, whereas binding of γ-aminobutyric acid (GABA) to inhibitory synapses inhibits an action potential. Glutamate is the most abundant neurotransmitter in the hippocampus, the area of the brain responsible for learning, emotions, and memory, and one of the main regions of the brain affected by AD. Loss of synapses is a major hallmark of AD, so determining the mechanisms leading to this loss will help in the understanding of AD pathophysiology and development of therapeutic targets.
Synaptic dysfunction
Mounting evidence suggests that AD is predominantly a disease of synaptic dysfunction and affects the ways in which neurons communicate with one another. Helping drive our understanding of these neuropathological changes in AD is Professor Rafael Luján at Castilla-La Mancha University, Spain. Using sensitive cutting-edge immuno-electron microscopy techniques alongside biochemical techniques, Luján investigated four key proteins associated with glutamatergic synapses: GIRK, mGlu5, AMPA and GABAB. Using AD mouse models, he has determined when and how these proteins are altered in the brain during disease. Pin-pointing the precise subcellular localisation has important implications for the function of these proteins and compromises the basic capability of neurons to process and transmit signals.
Mounting evidence suggests that AD is predominantly a disease of synaptic dysfunction and affects the ways in which neurons communicate with one another.
GIRK channels
GIRK channels may consist of four subunits (GIRK1, GIRK2, GIRK3, GIRK4), however in the brain these channels typically consist of GIRK1 and GIRK2 subunits. Luján’s research team employed technically challenging immuno-electron microscopy experiments to carefully examine the localisation of GIRK2 channels, which are important inhibitors of neuron activity and areprevalent in the hippocampus. Expression of GIRK2 was significantly reduced in the hippocampus of ten-month-old P301S mice (a mouse model of AD), compared with control mice. Such reduction of GIRK2 channels along the surface of these neurons and their redistribution, may alter pyramidal cell excitability leading to impaired cognitive function associated with AD.

MGlu5 receptors
In another recent study, Luján used innovative high-resolution immunoelectron microscopic analysis to investigate the expression and exact location of a second protein – mGlu5 – in a mouse model of AD. mGlu5 receptor plays an important role in slowing down synaptic communication in the brain, exerting this effect through associated G-proteins and second messengers. Expression of mGlu5 in the hippocampus of APP/PS1 mice with advanced AD was similar to the expression found in control mice. However, the APP/PS1 mice were found to have significantly lower mGlu5 levels along the neuronal surface of specific hippocampal neurons: the CA1 pyramidal cells and DG granule cells. These neurons have an important role in learning, and spatial and episodic memory. Conversely, the amount of mGlu5 found inside these neurons was higher in the AD mouse models. As Luján explains, ‘this shows for the first time that mGlu5 all over the plasma membrane in APP/PS1 mice may be a contributing factor to the memory deficits with severe synapse loss that accompany this AD model’.

AMPA receptors
The team studied synaptic AMPARs (glutamate receptors and cation channels) which are vital for synaptic transmission. They used immunolocalisation and immunoelectron microscopy methods to investigate AMPAR expression and subcellular distribution in tissue from APP/PS1 mice. Notably, levels of these proteins were measured in AD mice models of varied age and disease progression. Young APP/PS1 mice had a similar overall amount of AMPAR, and a similar subcellular expression in the hippocampus as control mice. However, the AD mice showed an age-dependent drop in AMPAR density at the synapses of both pyramidal cells and interneurons, compared to control mice. A reduction of AMPARs in the spines of dendrites was linked to a significant increase in AMPAR subunit proteins in specific cellular compartments. This age-dependent reduction of synaptic AMPARs in APP/PS1 mice might well correlate to impaired learning and memory seen in late-stage AD.

GABAB receptors
GABAB receptors formed by the subunits, GABAB1 and GABAB2 are abundant at excitatory synapses in the hippocampus. GABA receptors respond to the neurotransmitter γ-aminobutyric acid and help control activity and information processing in the hippocampus by regulating neuronal excitability and synaptic transmission. Luján and collaborators were the first to identify a reduction in plasma membrane GABAB receptors in CA1 pyramidal cells of the hippocampus in the mouse model of AD (APP/PS1), compared to controls. In parallel with this reduction was an increase in intracellular quantities of GABAB receptors subunits, and these findings were found to be associated with age.

Furthermore, the study also revealed a reduction in GABAB receptors in the axon terminals which contact CA1 pyramidal cells. These results are significant, and the team suggest such a reduction of GABAB receptors may lead to alteration of the GABAB mediated synaptic transmission in AD.

A recent study published by the team in the International Journal of Molecular Sciences used high-resolution immunoelectron microscopic analysis to investigate GABAB receptor density and localisation in granule cells in the dentate gyrus (DG) of the APP/PS1 mouse model of AD. The DG is important in the formation of memory and exploration of different environments. Study results showed decreased density of GABAB receptors on the neuronal surface in the postsynaptic compartments of granule cells in the APP/PS1 mice compared to controls. This reduction was paralleled by increases in cytoplasmic levels of GABAB1 receptor subunits. The finding of decreased GABAB receptors in the plasma membrane of axon terminals which contact granule cells’ dendritic spines, demonstrates presynaptic receptor changes in GABAB receptor density in addition to the postsynaptic changes already described. These intriguing findings verify the idea that GABAB -mediated modulation of excitability and synaptic transmission is altered and may well contribute to AD cognitive dysfunction.
Luján found changes in the number, distribution, and density of four key proteins in Alzheimer’s disease.
Luján’s innovative research has pin-pointed the progressive changes in the number, distribution, and density of four key proteins important for excitatory synapse function in AD. This offers important insight into our understanding of the mechanisms of the disease and has far-reaching consequences for individuals, families, and wider public health.

Personal Response
What are the next steps in your research? How might your findings be translated into results for patients and their families?
There are many other proteins associated with both excitatory and inhibitory synapses that need to be studied to define the molecular and cellular mechanisms underlying pathological conditions in neuronal signalling, including Alzheimer´s disease. The next steps include studying the involvement of these synaptic proteins in pathological conditions, as a first stage in identifying new molecular targets that in the future could have a therapeutic use in clinic.