Personalised strategies for cochlear implant surgery and hearing rehabilitation
Hearing loss is a common problem that usually develops with older age, affecting communication and making everyday life extremely difficult. If left untreated, it can lead to memory loss, social isolation, and mental health problems. Cochlear implants (CI) are an effective solution where hearing aids can’t help, but they are not fitted as often as they could be, as clinicians are wary of the perceived complications of surgical solutions and unaware how straightforward the procedure can be. Dr Aarno Dietz and his team at Kuopio University Hospital, Finland, are working on changing the game for CI surgery and hearing rehabilitation more broadly, through new evidence-based clinical strategies and patient-centred approaches.
Hearing loss is defined by a hearing threshold of 20dB in both ears, making it impossible for those affected to take part in normal conversation (typically around 60dB). It is a condition often associated with older age, meaning that the continuous increase in life expectancy in developed countries will make the problem increasingly common. Hearing loss can be sensorineural (related to the damage of sound receptors in the ear) or conductive (to do with chronic ear diseases or acquired stiffness of the auditory ossicles). As well as ageing, sensorineural hearing loss can be caused by factors such as excessive exposure to high levels of noise – constant use of earphones, or even sporadic standing under the speakers at a rock concert, for example.
Dealing with hearing loss is very important for the affected individual as it can severely impact their wellbeing and quality of life. Difficulties with everyday communication can cause embarrassment and loss of social interactions, and hearing loss can directly affect physical and mental health by causing anxiety, depression and, if left untreated for long, even cognitive decline. In cases of sensorineural hearing loss there are a few treatment options available including digital hearing aids, and – where hearing aids can’t provide adequate speech recognition in noise – cochlear implants (CI).

Cochlear implants
CIs are electronic devices which are surgically implanted into the temporal bone, in the side of the skull. It has two main elements: an external part or sound processor, which is usually worn behind the ear, and an internal body which is the implant itself. The internal body is surgically implanted behind the ear into a drilled bone bed in the temporal bone – it has an electrode array which is inserted into the cochlea. Sounds are picked up with the external sound processor and the sound is transformed to electric signals which are transmitted through the skin to the internal body. The implant body generates electric pulses, which directly stimulate the cochlear (hearing) nerve and in this way bypass the damaged hair cells in the inner ear. The electric signals provided directly to cochlear nerves finally carry the information via the primary auditory pathways to the brain.
Improving cochlear implant surgery
CIs are fitted through a surgical procedure which nowadays can be performed safely and comfortably under local anaesthesia, meaning more elderly people than ever could now be treated by the procedure. However, even in high-income countries, rates of CI surgery remain low (only up to 15% of people eligible for treatment), leaving too many people struggling with curable hearing loss. Adjunct Professor Aarno Dietz and his team at Kuopio University in Finland are taking a variety of approaches to making hearing rehabilitation more effective and more predictable. In particular, they aim to raise awareness of CI surgery among clinicians and the public, in an attempt to dispel the myth that the procedure is complicated or involved with too many surgical complications. ‘Since we have been developing CI surgery under local anaesthesia, there is no excuse anymore for not providing CIs to elderly people,’ says Dietz.
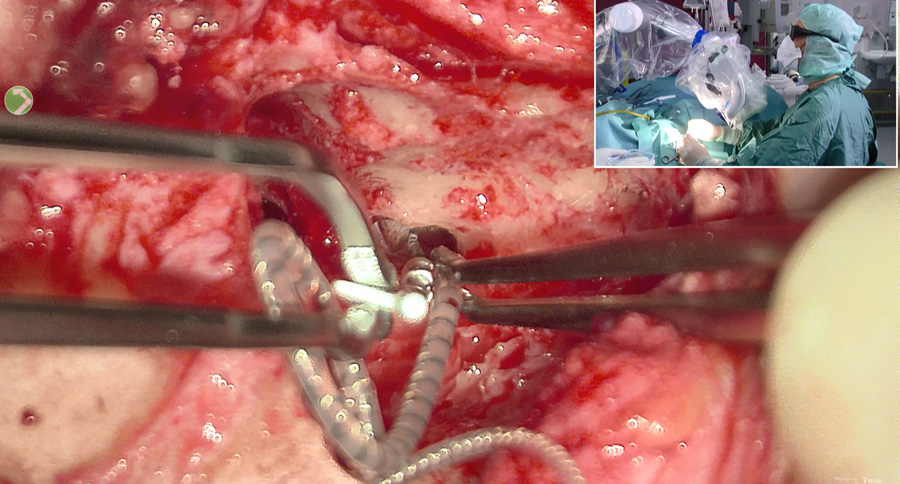
Both self-reported and hearing assessment tests are necessary to form a more comprehensive view of the outcome of CI treatment.To demonstrate the safety and benefits of CI, Dietz and his collaborator Professor Thomas Lenarz from the Hannover Medical School, Chair of one of Europe’s leading CI centres, recently conducted a study on patients who underwent the procedure under local anaesthesia. During the procedures the surgical team had to establish flawless communication with their patients, which they achieved with contra-lateral ear hearing (through the unaffected ear), visual, or haptic (touch) aids as needed. CI surgery was performed using the local anaesthetic lidocaine. In patients with residual hearing, the surgical team could monitor their hearing while inserting the CI electrodes so they could modify their actions according to the patient’s feedback. In this way, they could insert the electrodes without damaging delicate inner ear structures to give the best postoperative hearing outcomes. During the procedure, patients could already comment on loudness and pitch perception during the electrophysiological measurements. In comparison with surgery under general anaesthesia, the study patients could be directly referred to the ward without any need for surveillance in the wake-up room.

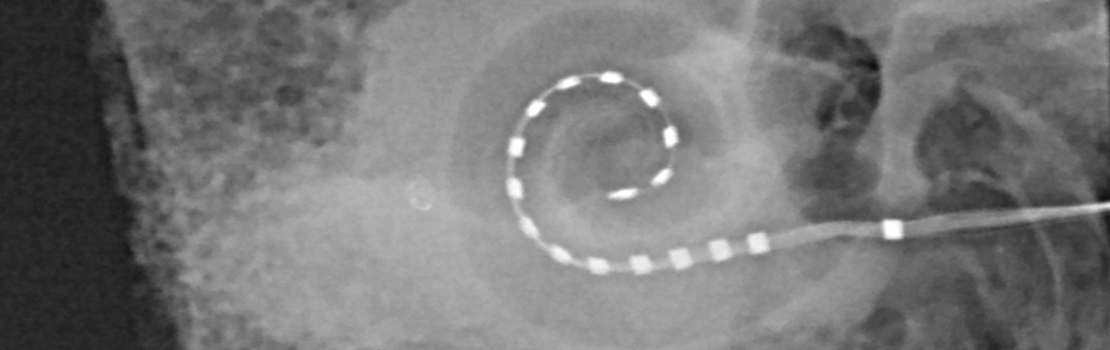
‘We were surprised by the positive results of our study; not only about the surgical results but in particular about patients’ acceptance to surgery under local anaesthesia,’ says Dietz. None of the patients needed a general anaesthetic to complete the procedure, and of the 71% who returned questionnaires, 97% were happy with the overall experience and 84% would choose the procedure under local anaesthesia again if given the option. There were 27 patients in the study with residual hearing and eligible for electric-acoustic stimulation who had the procedure performed under continuous monitoring and adjustment of the position of the electrode arrays according to their real-time feedback. ‘In these patients we used the concept of partial insertion, which was first described by my collaborator Professor Lenarz,’ says Dietz. ’This means we do detailed individual surgical planning, taking into account each patient’s individual inner ear size and preoperative hearing level, so we can choose the optimal electrode array length and determine how deep this particular electrode has to be inserted for best possible hearing preservation results.’ On the day after the surgery the team tested the patients’ hearing thresholds, and the results revealed the procedure had been successful for all of them.
Patient-centred outcome reporting
Despite this success, it is not always easy to predict how successful CI will be for the individual patient. This is a challenge that calls for a more integrated approach to understanding surgery outcomes, rather than just performing sound tests in a silent room. To tackle this, Dietz and his team designed another study, this time intended to explore the outcomes of the CI procedure from the patients’ perspective, including understanding more about their everyday hearing challenges.
Since the ability to understand speech, especially in noisy environments, plays a fundamental role in human communication and social interaction, the team decided to test this ability using the performance-based speech-in-noise (SIN) test, which assesses a patient’s hearing in the presence of competing sounds. As in the previous study, patients were also given questionnaires about the implants and the outcomes of their procedure before and after surgery, to gather what’s known as ‘patient-reported outcomes measures’.
The patients were followed up six months and one year after the surgery. The results showed compelling improvements in average hearing on all questionnaires, including the SIN tests at both follow-ups. The team also found that the lower the scores before the procedure, the larger the improvement in hearing afterwards. Interestingly, there was no association of the improvement shown on the questionnaires with the one revealed on the SIN tests, so the researchers concluded that the two approaches reveal different aspects of benefits from CI surgery. These results suggest that both types of tests (self-reported and sound assessments) are necessary to form a more comprehensive view of the outcome of the treatment and should therefore always be implemented in the clinics.
Dietz and colleagues are looking to advance precision medicine in hearing rehabilitation to achieve optimal results for each individual patient.Towards better hearing rehab
As part of his work to make hearing rehabilitation more effective and more predictable, Dietz and his co-researchers at the Kuopio Cochlear Implant Research group have started the ‘Towards Better Hearing’ project, an initiative that aims to improve hearing rehabilitation with hearing aids and implantable hearing devices in Finland through new, evidence-based clinical strategies.
These include the development of more sensitive, reliable, and cost-effective hearing tests for auditory screening, as well as developing tools to monitor the results of interventions. One recent development, which helps to build a profile of patients’ hearing, is the Finnish Matrix Test – the first sentence-speech intelligibility test in noise for the Finnish language. Recent advances in radiology, including virtual reality, for the evaluation of patients’ bone structures have also enabled the group to develop state-of-the-art imaging techniques that help them plan surgeries tailored to the individual’s needs. The team is currently running studies to test these techniques with a focus on causing as little damage as possible when implanting the devices, to preserve any residual hearing.
Dietz and colleagues are also improving hearing-related quality of life for patients by looking beyond the numbers and one-size-fits-all approaches, to advance precision medicine and achieve optimal results for each individual patient. Their ongoing research in this area is looking at new CI electrode designs and surgical techniques to make surgery even safer. Dietz is also collaborating with the Nordic-UK BrainNetwork, based at the University of Eastern Finland, to investigate the relationship between hearing rehabilitation and the prevention of cognitive decline in the form of diseases such as dementia.
Personal Response
The relationship between untreated hearing loss and cognitive impairment is a very interesting one, especially since both conditions usually develop with age. How is your current research approaching this relationship and what new information are you hoping it will bring to light?Hearing loss is a condition with a much broader impact than just the inner ear. We often think of it in a too simplistic way, in terms of impaired cochlear sensitivity to quiet sounds – basically what we see on the audiogram. However, hearling loss disability is mainly due to impaired speech perception in noise, which is the ability to segregate speech from competing noise. This is vital for normal daily life and requires intact auditory and central processing. Unfortunately, standard treatment with hearing aids often does not include control of the rehabilitation outcomes (ie, measuring speech perception in noise), leading to suboptimal results, analogous to treating hypertension with no regular blood pressure checks to monitor effects. Thus, patients unable to utilise acoustic amplification who would be eligible for cochlear implants, often go undetected for years. Therefore, we need more research on valid outcome measures which also include auditory processing and cognitive factors.