Stem cell-based cartilage regeneration technology
Harnessing the regeneration ability of stem cells, researchers are making strides in the treatment of large cartilage defects of the knee. A collaborative team including Dr Khay-Yong Saw, Dr Reza Ng, Dr Caroline Jee (Kuala Lumpur Sports Medicine Centre, Malaysia), and Dr Adam Anz (Andrews Research and Education Foundation, Florida, USA) demonstrate the safety and efficacy of peripheral blood stem cell (PBSC) injections for treating damaged articular cartilage in a recently published clinical trial. Their results showcase the potential for this innovative technology to meet a currently unmet medical need for many patients across the world suffering from joint damage.
Articular cartilage is the smooth, white tissue that covers the ends of our bones at the points where they come together to form joints. A highly specialised form of connective tissue, articular cartilage provides a smooth and lubricated surface for joints including the wrists, elbows, shoulders, hips, knees, and ankles. This slippery, shock-absorbing layer of tissue helps our bones to move against each other easily and with very little friction. Without articular cartilage, movement would be painful and difficult.
Unfortunately, articular cartilage is particularly susceptible to damage, either through injury or normal wear and tear. Cartilage is much softer and more flexible than bone, and not very good at healing itself; it has almost no regenerative potential in adulthood. Consequently, once articular cartilage is injured or gone there are very limited treatment options for patients.

This is where innovative research from Dr Khay-Yong Saw, Dr Reza Ng, and Dr Caroline Jee (Kuala Lumpur Sports Medicine Centre), and Dr Adam Anz (Andrews Institute, Florida) comes in. Years of research, including a recently published clinical trial, shows that peripheral blood stem cell injections offer exciting potential as an effective way of treating major articular cartilage damage.
This innovative technology has been developed to enable high-quality repair and regeneration of articular cartilage.
Cartilage defects
Articular cartilage damage can lead to chronic and painful symptoms including stiffness and swelling. Moving those joints becomes very painful and this damage can lead to arthritis later in life. There are limited surgical options available for people with damaged articular cartilage. The surgical goal is to stimulate the growth of new hyaline cartilage, the specialised tissue on the joint surface. One approach, known as microfracture, attempts to encourage the growth of new articular cartilage via the creation of a new blood supply. This involves using a sharp tool to make holes in the joint surface, which brings a new blood supply, new cells, and stimulates a healing response. The same outcome is sometimes achieved through drilling or abrasion arthroplasty. Another method known as autologous chondrocyte implantation involves new cartilage cells being grown in a laboratory and then implanted back onto the joint. Some patients will also have cartilage transferred from a healthy non-weightbearing part of the bone and have it grafted onto the defective area of the joint.
These methods all have their limitations, however, and fully repairing articular cartilage remains a significant unmet medical need. Consequently, many patients experience long and painful periods before undergoing a total joint replacement.
The power of stem cells
Building on many years of research, a pioneering approach involving stem cell technology has been developed by Saw, Ng, Jee, and Anz. Their goal? To place stem cells at the damaged area of the joint to stimulate cartilage growth (called chondrogenesis).
As Jee explains, ‘the science, surgical technique, harvesting, and storage of the peripheral blood stem cells and the postoperative rehabilitation programme that encompasses this innovative technology have been developed to enable high quality regeneration and repair’.

A stem cell is an immature, basic cell that has yet to specialise, or develop, meaning it has the potential to become any of the many types of cells in our bodies. For example, an undifferentiated stem cell may become a nerve, liver, heart, or blood cell, depending on the conditions they develop under. This of course means they have incredible regenerative potential.
The team’s newly developed stem cell technology is called ‘KART’ (KLSMC Articular Regeneration Technology). The aim is to achieve chondrogenesis by exploiting the natural healing qualities of a patient’s own peripheral blood stem cells (PBSC). Formed in the bone marrow, PBSC are the precursor of all the different types of blood cell. Early work carried out by Saw and colleagues identified PBSC as the stem cell of choice. Not only are they easily and safely harvested, but the researchers’ studies in animal models demonstrated their effective cartilage healing potential. Notably, PBSC can be cryopreserved for later use.

Cartilage regeneration
Following their successful findings in animal models, Saw and colleagues carried out a series of studies in humans to find out if their damaged knee joints could be restored using their KART technology. The researchers accessed the damaged knees using arthroscopic subchondral drilling (into the layer of bone just beneath the cartilage), a long-standing method of stimulating the bone marrow.
Subsequent (postoperative) intra-articular injections of PBSC plus hyaluronic acid (HA) were then carried out. HA is a naturally occurring substance found in the body’s connective tissues with proven ability to improve cartilage health and repair, speed up the development of immature cells into chondrocytes, and reduce joint inflammation.
These studies highlighted the potential of their method for the successful regeneration of damaged articular cartilage. Not only could their procedure effectively and simultaneously treat multiple lesions, it was also shown to treat ‘kissing’ bone-on-bone lesions (not rare but serious injuries where there is damaged tissue on both sides of the bone joint).
Notably, more than 70% of patients with ‘unmet medical needs’ who received the articular regeneration treatment showed significant improvement.
A successful clinical trial
On the basis of the team’s earlier clinical studies, a larger clinical trial was conducted to establish whether PBSC regeneration therapy could improve the functional outcomes for patients and reduce their knee pain. PBSC therapy was compared to patients receiving traditional treatments (of HA injections and physiotherapy). Patients with large cartilage knee defects (classified as ≥3cm2) were recruited to a 24-month dual-centre, open-label phase IIB randomised controlled trial (RCT). A group of 69 patients were studied over the course of two years, with half randomly receiving HA and PBSC injections following arthroscopic subchondral drilling, and the other half receiving the HA injections to act as a control group. Both patient groups also received post-treatment physiotherapy.

After 24 months, the patients’ knee pain was scored, and their rate of cartilage repair was recorded. Primary outcome measures included knee function, measured using standardised IKDC scores (a knee-specific measure of symptoms, function, and sports activity). Pain reduction was also measured, using KOOS scores (which assess the health, symptoms, and functionality of the knee).
As a secondary outcome measure, the researchers used the ‘MOCART’ score (Magnetic resonance Observation of Cartilage Repair Tissue) as a way to assess chondrogenesis. MRI scans revealed there were no unexpected or adverse events throughout the duration of the study.
The interim analysis yielded promising results. After just six months following articular regeneration treatment, patients showed statistical improvement in pain scores compared to the HA control group. Only 12 months following treatment, the regeneration treatment group showed significant improvement across all measures: knee pain, knee function, and cartilage repair. Notably, more than 70% of patients who received the articular regeneration treatment showed significant improvement.
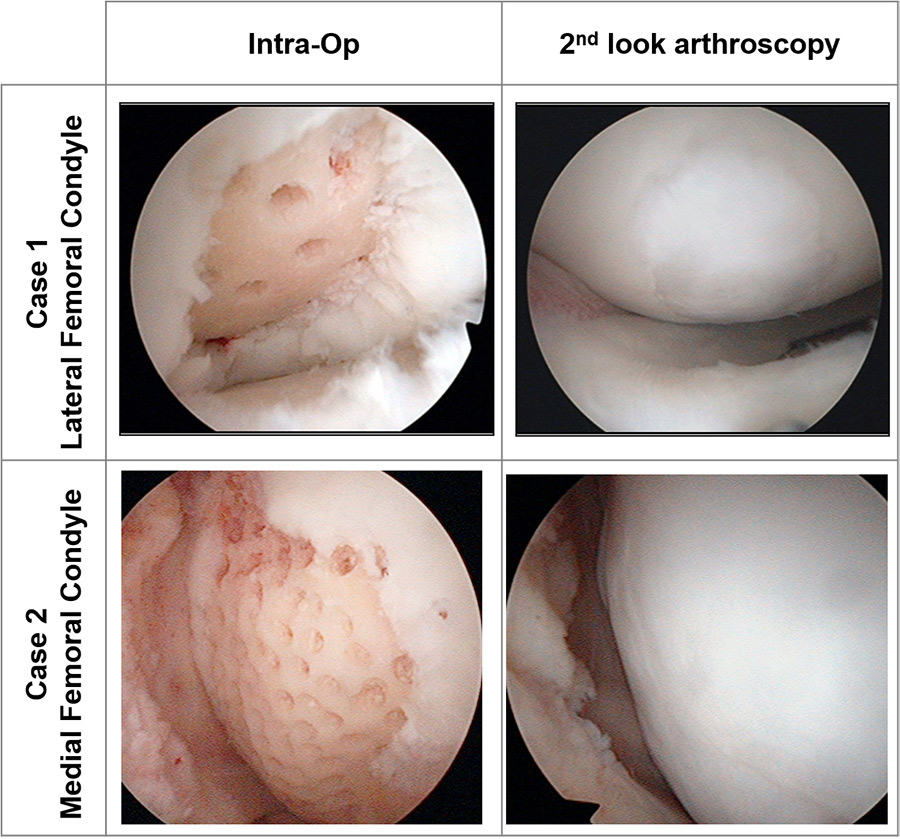
Right panels: Arthroscopic images at two years following surgery, showing fully regenerated cartilage.
The potential of regeneration therapy
Articular regeneration treatment offers a safe and highly effective way of treating patients with massive cartilage defects. The researchers are now aiming to proceed to a Phase 3 trial in 2022. Excitingly, the research team believe that their regeneration technology could be applicable to other joints.
Personal Response
How long do you think it will take for articular regeneration treatment to be the ‘go-to’ treatment for cartilage injury?
This will take at least the next five years following the completion of the multi-centre Phase 3 RCT with subsequent regulatory approval. The process starts with the training of orthopaedic surgeons and familiarising the treating surgeons on the principles of chondrogenesis with specialised surgical techniques and multiple post-operative PBSC injections. Educating the treating physiotherapists on the importance of post-operative weightbearing exercises with movements of the operated joints is also an essential component. PBSC harvesting centres need to be established for cell preparations, cryopreservation, storage, and delivery.