Bacterial biofilms: A persisting public health challenge
Foodborne and waterborne diseases are major global health threats, exacerbated by the ability of bacteria to gain new characteristics, constantly evolve towards increased fitness in their environment, and by developing resilience through biofilm formation. Utilising emerging technologies such as elevated hydrostatic pressure and application of novel bacteriocin and bactericidal compounds, Professor Aliyar Cyrus Fouladkhah and his team in the Public Health Microbiology laboratory at Tennessee State University are mitigating the risk of these infectious diseases and bacterial biofilms.
Bacterial diseases are a major public health challenge in the United States and worldwide, especially those infectious diseases associated with water and food supplies. While much research has focused on bacteria that are found living freely in the environment, less work has been done to understand the ecology of bacteria who can form sessile communities in the form of biofilm. Biofilms are closely packed communities of cells that settle on living or inert surfaces and surround themselves with secreted exopolysaccharides (complex sugars), essentially creating a miniature ‘city for microbes’. Living within a biofilm community allows the bacteria to interact and communicate in ways that free-living bacteria (planktonic cells) are unable to, for example by passing biochemical signals and exchanging genetic information.
Prof Aliyar Fouladkhah and his research group at Tennessee State University aim to understand more about these biofilm-forming bacteria. In doing so, they hope to encourage a preventative approach which reduces the disease-causing potential, or pathogenicity, of the bacteria and their prevalence in food and water supplies before they are able to cause infections. Prof Fouladkhah’s research has focused on the use of emerging interventions such as elevated hydrostatic pressure and application of novel bacteriocin and bactericidal compounds to achieve this, particularly targeting Shiga toxin-producing Escherichia coli (a form of E. coli that causes foodborne disease and a potentially fatal kidney complication called Hemolytic Uremic Syndrome), non-typhoidal Salmonella serovars, public health-significant serovars of Listeria monocytogenes, and pathogenic species of Cronobacter. The term ‘serovar’ is used to identify specific subgroups of bacteria within a species of bacteria.
Bacterial biofilms are a notable public health challenge. Their understanding is important for prevention of infectious diseases associated with food and water supplies
Prof Fouladkhah works closely with domestic and global stakeholders in a holistic approach for dissemination of his applied research deliverables to assure microbiological safety, regulatory compliance, and economic feasibility for the reduction of these foodborne pathogens. Prof Fouladkhah’s research group has provided extramurally funded education opportunities for undergraduate, Master’s and PhD students, post-doctoral and visiting scholar fellows and, in addition, has held public health workshops, training events, certification programmes, and consultations in the Dominican Republic, Guatemala, South Africa, and Haiti.
Biofilm formation of bacterial pathogens
Since the historic outbreak of cholera near Broad Street in London in 1854, safety of water supplies had been an integral part of public health practices and infrastructure. Prof Fouladkhah and his team investigated the persistence of several bacteria in surface water – Listeria monocytogenes, Shiga toxin-producing Escherichia coli and Salmonella serovars stored for 28 days at either 5, 25 or 37oC. The results of the study demonstrate that these microorganisms are able to survive under low-nutrient conditions and highlight the need for measures to prevent initial contamination of food and water sources. For example, agricultural crops may come into contact with surface water before or after they have been harvested, presenting a potential contamination route in their processing. Many raw agricultural commodities will not undergo any additional “kill step” by the consumer to eliminate microbial pathogens, so the contamination of these crops is an important route of transmission of infectious diseases to consumers. The formation of biofilms has also been associated with transfer and enhanced establishment of crop pathogens, providing further support for the need to increase understanding of biofilm-producing bacteria and the methods to prevent their formation and proliferation.
Biofilm formation and inactivation of Shiga toxin-producing E. coli
Out of more than 700 recorded waterborne disease outbreaks in the United States between 2009 and 2017, at least 15 were caused by E. coli and/or Salmonella. During this same period, 10,596 episodes of illness were related to infections with E. coli, including over 2,000 episodes of hospitalisation and almost 4,000 deaths. Additional data show that in samples of untreated surface water collected from 27 sites in North America, around 80% of samples contained human pathogens, such as E. coli.
Prof Fouladkhah suggests that in addition to persistence in water after 28 days, the pathogenicity of the bacteria may remain, meaning that they do not become significantly less harmful the longer they remain in surface water. Further experimental studies are needed to investigate the expression of virulence-associated genes in E. coli, as well as
L. monocytogenes and Salmonella serovars using culture-independent methods such as reverse transcription PCR or in vitro pathogenicity assays. The term virulence describes the ability of a pathogen or microbe to infect or cause disease in a host.
The conclusions of the research done by the group at Public Health Microbiology laboratory show that these microorganisms are able to survive for prolonged periods of time, even in low-nutrient environments, thus highlighting the need to prevent contamination of food and water sources in the first place. The bacterial pathogen investigated were also capable of forming bacterial biofilms on abiotic surfaces, further highlighting the needs for preventive approaches such as the recently implemented U.S. Food Safety Modernization Act.
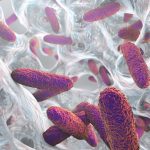
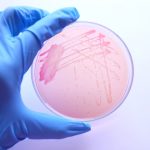
Pressure-stressed, multi-drug resistant, and susceptible Salmonella serovars
Due to advancements in technologies, elevated hydrostatic pressure is being increasingly used by the food and pharmaceutical industry to prevent contamination with pathogenic organisms and those with the potential to spoil the food product. The process of elevated hydrostatic pressure (or pascalisation) involves placing products in sealed packaging and then putting them in an airtight steel compartment containing water. Pumps are then used to create a high-pressure environment within this compartment, while ensuring there is no change in temperature that could affect the quality of the food product. Most microorganisms are unable to survive the elevated pressure, thus ensuring the safety of the product. However, there is less evidence for what happens to the pathogens after exposure to ultrahigh pressure, i.e. what happens to those bacteria that manage to survive?
Prof Fouladkhah’s research group was one of the first to publish research on this subject. They compared the ability of pressure-stressed and wild-type (normal) Salmonella serovars to produce biofilms. The findings of his study showed that both forms of Salmonella had similar biofilm-producing capabilities. This is important as it increases understanding of how stressors, such as pressure, do not always result in resistant bacteria with increased pathogenicity and that the same prevention strategies will target both pressure-stressed and wild-type phenotypes of Salmonella serovars. Previously conducted research had also shown that multi-drug resistant and susceptible Salmonella serovars could form bacterial biofilm that are more resistant to common antimicrobial intervention when compared to planktonic cells.
Biofilm and planktonic cells of bacterial pathogens are climate-sensitive infectious diseases and their prevalence and proliferation will be augmented under the landscape of climate change.
Cronobacter sakazakii in infant care settings
C. sakazakii is a bacterium that can cause infection in a range of age groups, but perhaps the biggest threat is its ability to persist in dry conditions, such as infant formula, and the subsequent risk of fatal infections in premature or very young infants.
Prof Fouladkhah’s group has documented many historic global outbreaks of C. sakazakii to help understand the epidemiology behind infections with this pathogen. He also discusses measures that could be taken to reduce the risk of infants becoming infected, including encouraging breastfeeding and best practices of preparation of infant formula as dictated by regulatory and public health agencies. In addition to direct measures, there are measures which could be implemented in the production phase, for prevention of contamination of infant products with this opportunistic pathogen.
Producers need to be aware of the limitations of sampling small quantities of their food products. Since the selected sample may not show contamination, relying on sampling and testing alone could provide false sense of security about the safety of the product. There are a number of methods which can be used to decrease the chance of contamination from
C. sakazakii, such as pressure or temperature-based interventions and use of disinfectants to prevent biofilm formation in food contact surfaces of production and preparing areas. Rather than relying on testing methods alone, producers can be guided by holistic and preventive-based food safety management systems such as Hazard Analysis Critical Control Point and those articulated under the U.S. Food Safety Modernization Act.

Control and biofilm formation of Listeria monocytogenes
Listeria monocytogenes is in the top three causes of foodborne death episodes in the U.S. It is also one of a small number of foodborne pathogens which can multiply under refrigeration conditions, making it major threat to the food industry and for public health. Previous outbreaks of this pathogen have had significant impacts upon consumer security and resulted in economic losses for stakeholders. Furthermore, L. monocytogenes can persist under other adverse conditions, such as low pH or high salt concentrations. Pregnant women, the very young, elderly, and immunocompromised individuals are more susceptible to infections with this ubiquitous and opportunistic pathogen.
Therefore, there is a need to develop effective and safe methods to eliminate this bacterium. The Public Health Microbiology laboratory have been doing just that using a combination of mild heat and nisin (a naturally occurring antibiotic) to show that a synergistic approach does lead to inactivation of L. monocytogenes, but there is a tightly controlled trade-off between the use of nisin and increased treatment time of elevated temperature and pressure.
L. monocytogenes is particularly capable of forming biofilms on various biotic and abiotic surfaces, making its prevention and elimination in food supplies a daunting and complicated task.
Bacterial biofilms and foodborne diseases
Previous research has reviewed the impact that environmental stressors, such as dramatic changes in temperature, pressure or pH, can have on bacteria. Exposure to such stressors may cause the bacteria to become more resistant to them or the sensitivity of bacteria to that particular stressor may be enhanced. Prof Fouladkhah is interested in the use of emerging technologies such as elevated hydrostatic pressure and use of novel bacteriocin and bactericidal compounds for development of adoptable, microbiologically efficacious, and economically feasible approaches for control of planktonic cells and biofilms of foodborne pathogens of public health concern.
In his recent publications, Prof Fouladkhah also illustrated that microbial food safety and waterborne diseases will continue to remain important public health challenges in the landscape of climate change as increases in environmental temperature could play a very substantial role on the proliferation and spread of infectious diseases. He explains that extreme weather events may additionally promote the spread of microbial pathogens from animal food production facilities to surface water. In these ways, climate change will be a ‘threat multiplier’ for food and waterborne infections. There is a critical need to develop legislation and evidence-based best practices to ensure the safety of food and water supplies in the coming years.
Personal Response
What do you think is the most effective way to assure safety of our food and water supplies?